
Introduction
Metal components fail not from the metal itself, but from surface degradation. Corrosion, wear, friction, and heat attack the outer layer first, forcing unplanned shutdowns and replacement cycles that compound costs fast.
The right surface finishing treatment determines how long components last, how they perform under stress, and total lifecycle maintenance costs. According to NACE International (now AMPP), corrosion alone costs the global economy $2.5 trillion annually—3.4% of global GDP. In gas pipelines, corrosion drives 25.7% of all incidents, surpassing external interference as the leading failure cause.
Choosing the wrong process means paying those costs. This guide covers 8 metal surface finishing treatments: how each works, where it performs best, and what trade-offs to expect.
TLDR
- Surface finishing modifies metal outer layers to improve corrosion resistance, hardness, wear resistance, or aesthetics
- The 8 main processes: electroplating, electroless plating, powder coating, anodizing, passivation, diffusion coatings, heat treatment, and mechanical finishing
- Selection depends on base metal, operating environment, performance requirements, and budget
- Diffusion coatings (boronizing, aluminizing) outperform most alternatives in extreme wear and corrosion conditions
- Match treatment to your environment early — wrong specs lead to costly rework and premature failure
What Is Metal Surface Finishing?
Metal surface finishing is any process that alters or coats the outer layer of a metal part to enhance corrosion resistance, surface hardness, wear resistance, conductivity, adhesion, or appearance.
Three broad categories define surface finishing methods:
- Mechanical processes physically modify the surface (shot peening, abrasive blasting)
- Coating/plating processes add a material layer (electroplating, powder coating, anodizing)
- Thermochemical/diffusion processes change surface composition at a molecular level (boronizing, carburizing, nitriding)

Choosing the right process — not just any treatment — determines how long a part lasts under real operating conditions. The eight processes covered in this guide represent the most widely used methods across industrial manufacturing today.
Why Metal Surface Treatment Matters in Industrial Applications
Untreated metal surfaces in demanding environments face premature wear, corrosion-driven failures, unplanned shutdowns, and replacement costs. The Federal Highway Administration estimates direct annual corrosion costs in the U.S. at $276 billion (3.1% of GDP).
In liquid pipelines, corrosion causes 26% of high-impact incidents affecting people and environment. Oil and gas production sees over 25% of safety incidents driven by corrosion-related failures from H₂S, CO₂, and oxygen exposure.
The right surface treatment delivers:
- Extended component life
- Reduced maintenance frequency
- Protection against chemical attack
- Use lower-cost base materials without sacrificing performance
The scale of potential savings makes the case plainly: the NACE IMPACT study notes that applying available corrosion control practices could yield $375 billion to $875 billion in global savings annually—15% to 35% of total corrosion costs. Choosing the right treatment process is where that value is either captured or left on the table.
The 8 Metal Surface Finishing Treatment Processes
Each process serves distinct performance needs — some excel at corrosion protection, others at hardness, aesthetics, or a combination. Understanding what each process actually does to the metal surface is what separates the right specification from an expensive rework.
Electroplating
Electroplating deposits a thin metal layer onto a substrate using electrical current passed through a liquid bath containing dissolved metal ions. The part acts as the cathode and attracts ions to its surface.
Common metals and applications:
| Metal | Standard | Typical Thickness | Primary Use |
|---|---|---|---|
| Zinc | ASTM B633 | 5–25 µm | Sacrificial corrosion protection for iron and steel |
| Hard Chromium | AMS 2460 | 2.5 to >25 µm | Extreme wear, abrasion, and fretting resistance |
| Nickel | ASTM B689 | 5–200 µm | Buildup for worn parts, underplating, corrosion barrier |
| Gold | MIL-DTL-45204 | 0.00005–0.0001" | Electrical contacts, conductivity, solderability |
Key strengths:
- Consistent coating thickness on flat/simple geometries
- Good corrosion protection
- Improved conductivity
- Decorative finishes
Critical limitation—hydrogen embrittlement:
High-strength steels (≥1000 MPa / 31 HRC) risk catastrophic delayed brittle failure from atomic hydrogen entering the steel lattice during plating. ASTM B850 and AMS 2759/9 mandate embrittlement-relief baking at 190–220°C for minimum 22 hours, starting within 1–3 hours after plating.
Other trade-offs:
- Limited coverage on complex internal geometries
- Uneven current distribution leaves blind holes vulnerable
- Requires strict bath chemistry control
Electroless Plating
Electroless plating (autocatalytic plating) coats substrates through chemical deposition without external electrical current. A self-catalyzing chemical reaction deposits metal uniformly regardless of part geometry.
Core advantage: Uniform coating on complex shapes, threads, recesses, and internal surfaces—electroless nickel plating (ENP) deposits equally across all areas in contact with solution because no current concentrates on edges.
Performance tuning via phosphorus content:
| Phosphorus Content | Microstructure | Hardness | Applications |
|---|---|---|---|
| Low (2–4% P) | Microcrystalline | 620–750 HK | Severe abrasion, mining, tooling |
| Medium (5–9% P) | Mixed | Balanced | General-purpose industrial machinery |
| High (>10% P) | Amorphous | Lower hardness, superior corrosion resistance | Oil & gas, marine, severe chemical environments |

High-phosphorus ENP eliminates grain boundaries, providing superior salt-spray and acid resistance ideal for severe corrosive environments.
Best-suited applications:
- Precision components
- Intricate geometries
- Parts requiring tight dimensional tolerance after coating
- Valves, manifolds, internal bores
Limitations:
- Slower deposition rates than electroplating
- More sensitive bath maintenance
Powder Coating
Powder coating electrostatically applies dry polymer powder to a metal surface, followed by oven curing to produce a hard, protective polymer finish—thicker and more impact-resistant than liquid paint.
Typical industrial powder coatings achieve 60 to 150 µm dry film thickness in single coats. The industrial and consumer goods sectors account for 24.3% of the global powder coatings market.
Temperature resistance by polymer chemistry:
- Standard thermosets (epoxy/polyester): ~200°C continuous service
- High-temperature silicones: Up to 538°C (1000°F) for engine exhausts, furnaces, agricultural equipment
Best-use applications:
- Architectural components
- Consumer products
- Outdoor equipment where aesthetics and moderate corrosion resistance are primary goals
Limitations:
- Not suitable for extreme temperature environments (standard formulations)
- Limited chemical resistance compared to metallic coatings
- Cannot protect tight-tolerance machined surfaces
Anodizing
Anodizing is an electrochemical process that converts the surface of aluminum (and some non-ferrous metals) into durable aluminum oxide. The layer grows from the base metal outward, integrating with the substrate rather than sitting on top.
MIL-A-8625F types for aluminum:
| Type | Acid | Thickness Range | Characteristics |
|---|---|---|---|
| Type II | Sulfuric | 1.8–25 µm | Conventional coating for general corrosion protection and dyeing |
| Type III (Hardcoat) | Sulfuric (low temp) | 12.7–114 µm | Heavy, dense, abrasion-resistant surface |
Type III hardcoat achieves 520–700 HV microhardness (roughly 50–60 HRC equivalent). Because the coating is aluminum oxide, it is exceptionally hard but brittle.
Key strengths:
- Excellent corrosion resistance for aluminum
- Improved surface hardness
- Allows color dyeing
- No coating adhesion issues (grows from base metal)
Core limitations:
- Primarily limited to aluminum and its alloys
- Coating can be brittle under impact, especially on sharp edges
- Titanium (AMS 2488) and magnesium (AMS 2478) anodizing exists but less common
Passivation
Passivation is a chemical treatment—using nitric or citric acid—that removes free iron and surface contaminants from stainless steel. Removing contaminants accelerates formation of a continuous chromium oxide passive layer.
Critical distinction: Passivation is not a coating. It removes microscopic surface contaminants and enhances the native oxide layer. It does not alter dimensions or surface hardness.
Where passivation is critical:
After machining, welding, or cutting stainless steel—these operations disturb the passive layer and introduce iron contamination. Passivation is mandatory in:
- Food processing (sanitary surfaces)
- Pharmaceutical manufacturing (ASME BPE standard)
- Medical devices (FDA recognizes ASTM F899 and ASTM F86)
Governing standards:
- ASTM A967: Specification for nitric/citric acid methods with verification tests
- AMS 2700: Aerospace equivalent for corrosion-resistant steels
- ASTM A380: Pre-cleaning, descaling, and pickling protocols
Limitation: Passivation enhances natural stainless steel properties but does not add hardness or wear resistance. It is a corrosion-specific treatment, not a mechanical performance upgrade.
Diffusion Coatings — Boronizing and Aluminizing
Diffusion coatings are thermochemical processes where boron or aluminum atoms are driven into the base metal surface at elevated temperatures via chemical vapor deposition. This forms an intermetallic compound that becomes part of the metal itself, not just a coating on top.
Fundamental difference from other treatments:
Because the coating is metallurgically bonded at a molecular level, it cannot peel, flake, or delaminate. This eliminates the risk of overlay spalling under severe mechanical stress or thermal cycling.
Boronizing—extreme hardness for wear resistance:
Boronizing diffuses boron into steel, creating an intermetallic boride layer (FeB or Fe₂B).
| Phase | Hardness (Vickers) | Characteristics |
|---|---|---|
| Fe₂B | 1400–1600 HV | Preferred industrially; tough and highly wear-resistant |
| FeB | 1900–2100 HV | Extremely hard but brittle; prone to microcracking |
| Tungsten Carbide (WC) | 700–2500 HV | Industry benchmark for cutting tools |
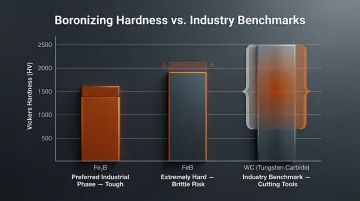
Boronizing allows inexpensive carbon steels to achieve surface hardnesses matching tungsten carbide. Oil and gas operations use boronized downhole tubing, slurry pumps, and valves to extend service life 3 to 10 times compared to untreated steels.
Aluminizing—high-temperature oxidation resistance:
Aluminizing diffuses aluminum into steel or nickel-based superalloys, forming a β-NiAl or iron-aluminide layer. At extreme temperatures, this layer forms a slow-growing, self-healing α-Al₂O₃ (alumina) scale that blocks oxygen and carbon diffusion.
Aerospace gas turbine blades and petrochemical ethane cracker coils rely on aluminizing. Case studies show upgrading to alumina-forming coils extends furnace run lengths from 33 days to over 400 days.
Industries where diffusion coatings deliver highest value:
- Oil and gas
- Petrochemical
- Mining
- Agriculture
- Aerospace
Service providers specializing in diffusion coatings — such as VaporKote — formulate custom powder mixes on-site and operate large-capacity industrial furnaces capable of processing components up to 68 inches in diameter, making the process practical for large industrial parts that other coating methods cannot accommodate.
Heat Treatment — Case Hardening (Carburizing and Nitriding)
Case hardening is a thermochemical diffusion process producing a hard, wear-resistant outer case while preserving a tough, ductile core. This gives a single part two different mechanical property zones.
Carburizing vs. nitriding:
| Metric | Gas Carburizing (AMS 2759/7) | Gas Nitriding (AMS 2759/6) |
|---|---|---|
| Process Temperature | 788–954°C (1450–1750°F) | 491–566°C (915–1050°F) |
| Typical Case Depth | 0.25 mm to >1.5 mm | Up to 0.88 mm (0.035") |
| Quench Required? | Yes (oil or gas) | No |
| Distortion Risk | High (phase change & thermal shock) | Very low |
| Primary Applications | Heavy-duty gears, camshafts, transmission components | Precision crankshafts, valves, tooling |

Carburizing diffuses carbon into low-carbon steel surfaces. It requires quenching, producing deeper case depths suited for heavy-load applications.
Nitriding diffuses nitrogen at lower temperatures. It operates below the tempering temperature with no quench, preserving tight tolerances for precision components. Parts can be finish-machined before nitriding, saving manufacturing costs.
Key limitation: Heat treatment modifies internal structure and can cause distortion if not controlled precisely. Parts with tight dimensional tolerances may require post-treatment grinding. The process is not reversible.
Mechanical Finishing — Shot Peening and Abrasive Blasting
Though both processes propel media at metal surfaces, their engineering purposes differ entirely. Abrasive blasting is a cleaning process; shot peening is a structural modification process.
Shot peening—engineering fatigue life:
Governed by AMS 2430 and SAE J443, shot peening bombards components with spherical media (steel, glass, ceramic). Kinetic energy causes localized plastic deformation, yielding compressive residual stress at the surface.
Because fatigue cracks require tensile stress to propagate, this induced compressive layer acts as a barrier. Studies show optimized shot peening increases the high-cycle axial fatigue limit of structural steels by 21.8%.
Process intensity is controlled using Almen strips to ensure repeatable compressive stress profiles.
Abrasive blasting—surface preparation:
Abrasive blasting uses angular or mixed media to remove rust, mill scale, and old coatings, creating a roughened surface profile (anchor pattern) necessary for coating adhesion.
ISO 8501-1 cleanliness grades (Sa designations):
- Sa 1 (Light Blast Cleaning): Removes poorly adhering mill scale, rust, paint
- Sa 2 (Thorough Blast Cleaning): Removes most mill scale and rust; residual contamination must be firmly adhering
- Sa 2.5 (Very Thorough Blast Cleaning): Near-white metal; traces may show as slight stains
- Sa 3 (Blast Cleaning to Visually Clean Steel): White metal; completely free of visible contaminants
Note: ST2 and ST3 designations refer to manual hand and power tool cleaning (scraping, wire brushing, grinding), not abrasive blasting.
How to Choose the Right Metal Surface Finishing Process
The right process is determined by operating environment and the failure mode you're solving for — not by cost alone or habit. What works for a decorative consumer product will actively harm a downhole oil drilling component.
Key selection factors:
- Base metal type and compatibility — Not all processes work on all metals (anodizing is aluminum-specific; passivation is stainless-specific)
- Primary failure mode — Wear, corrosion, fatigue, oxidation, or combination
- Operating temperature range — Standard powder coatings fail above 200°C; aluminizing protects to 1100°C
- Dimensional tolerance requirements — Carburizing causes distortion; nitriding and passivation preserve tolerances
- Part geometry — Electroless plating covers complex internal surfaces; electroplating struggles with blind holes
- Required service life vs. budget — In high-failure-cost environments, diffusion coatings or advanced case hardening typically deliver lower total cost of ownership than repeatedly replacing undertreated parts

Even with the right criteria, several patterns reliably lead to poor outcomes:
Common selection mistakes to avoid:
- Choosing a process based on familiarity rather than fit
- Specifying decorative or cosmetic finish when structural surface treatment is needed
- Overlooking compatibility between coating and substrate (for example, hard chrome on high-strength steels prone to hydrogen embrittlement)
- Underestimating post-treatment machining requirements for precision parts
Frequently Asked Questions
What is metal treatment?
Metal treatment encompasses any process used to modify surface or internal properties of metal—including heat treatment, surface coatings, plating, and chemical diffusion—to improve hardness, corrosion resistance, wear resistance, or other performance characteristics.
What are the main methods of metal surface treatment?
Metal treatment methods fall into three categories: mechanical processes (shot peening, abrasive blasting), coating and plating processes (electroplating, powder coating, anodizing, passivation), and thermochemical/diffusion processes (boronizing, aluminizing, carburizing, nitriding). Each category addresses different failure modes and performance needs.
What are ST2 and ST3 surface preparation standards?
ST2 and ST3 are ISO 8501-1 manual cleaning standards for steel surfaces. ST2 (thorough hand tool cleaning) removes loose mill scale, rust, and coatings. ST3 (very thorough hand tool cleaning) achieves a more uniform metallic sheen. Both are baseline surface preparation grades used before applying protective coatings.
What is the difference between electroplating and electroless plating?
Electroplating uses external electrical current to deposit metal ions onto a substrate, while electroless plating relies on a self-catalyzing chemical reaction without electricity. The key practical difference: electroless plating deposits uniformly on complex geometries and internal surfaces where electroplating cannot reach evenly.
Which metal surface treatment offers the best wear and corrosion resistance for heavy industrial use?
For extreme wear and corrosion in heavy industrial environments, diffusion coatings such as boronizing and aluminizing consistently outperform other surface treatments because the intermetallic compound formed is metallurgically bonded to the base metal, so it cannot peel or delaminate. Boronized surfaces achieve hardness exceeding tungsten carbide cutting tools.
How long do metal surface treatments last?
Service life depends on the process and operating environment. Decorative coatings like powder coat perform well in mild conditions but degrade quickly under heat or chemical exposure. Diffusion coatings and case hardening treatments can extend component life many times over untreated equivalents, sometimes saving hundreds of thousands of dollars annually in replacement and downtime costs.


