
Introduction
Metals used in oil drilling equipment, aerospace components, and agricultural machinery nearly always undergo heat treatment before reaching service. Petrochemical, mining, and manufacturing sectors depend on it to help components survive extreme wear, temperature swings, and corrosive environments.
Yet heat treatment is widely misunderstood as a single process, when it's actually a family of controlled thermal techniques. That distinction matters: poor material selection rooted in that misunderstanding leads to preventable component failures that cost industries millions in downtime and repairs.
According to industry data, the global heat treating market reached $109.74 billion in 2025 and is projected to grow to $164.3 billion by 2034, with approximately 80% of all processes applied to steel products. In the automotive sector alone, an hour of unplanned downtime in a large plant now costs $2.3 million—or more than $600 a second. This guide breaks down what heat treatment is, how each core process works, and how to choose the right method for your application.
TL;DR
- Heat treatment changes metal's internal structure and mechanical properties through controlled heating and cooling, leaving the part's geometry unchanged
- Outcomes depend on three variables: temperature, soak time, and cooling rate — adjust any one and the material properties shift
- Core methods include annealing, hardening, tempering, and diffusion treatments such as boronizing and carburizing
- Correctly applied, heat treatment extends part life and prevents premature failure in demanding industrial environments
What Is Heat Treatment?
Heat treatment is a group of controlled thermal processes—heating and cooling a metal to specific temperatures, holding it there for a defined time, and then cooling it—to intentionally alter its microstructure and thereby its physical and mechanical properties. The primary goal is changing material behavior, not shape.
That separates it from hot forming or welding, where heating is incidental to shaping. ASTM A941 explicitly states that heating for the sole purpose of hot working is excluded from the definition of heat treatment. Welding creates a Heat-Affected Zone with altered microstructures, but this is an uncontrolled byproduct—engineers typically specify Post-Weld Heat Treatment (PWHT) to correct the residual stresses and brittle microstructures it inadvertently creates.
Why Heat Treatment Remains Essential
Many metals in their raw or machined state lack the hardness, ductility, or wear resistance that demanding industrial environments require. As-cast and machined parts commonly retain:
- Internal residual stresses from prior forming or machining operations
- Uneven grain structures that concentrate stress under load
- Soft phases (such as ferrite) that wear quickly under abrasive friction
Heat treatment corrects all three without requiring a different base material.
For example, skipping or improperly executing PWHT leaves residual tensile stresses and hydrogen trapped in the metal, which has caused catastrophic hydrogen-induced cracking and pressure vessel failures in petrochemical operations.
How Does Heat Treatment Work?
Heat treatment operates through a defined sequence—heat the metal, hold it at temperature, then cool it in a controlled way. Each variable in this sequence directly determines the end properties of the part.
Heating and Soaking
The process begins when the metal is loaded into a furnace and heated to a target temperature, often called the transformation or critical temperature. At this point, the metal's internal crystal structure (microstructure) begins to change phase—typically shifting from one crystalline arrangement to another, such as ferrite to austenite in steel.
Once at temperature, the metal is held (soaked) for a period that allows heat to penetrate uniformly throughout the part. The industry standard rule of thumb calls for one hour of soak time per inch of cross-sectional thickness. This ensures adequate time to avoid thermal gradients and achieve complete austenite transformation. Thicker parts require longer soak times; insufficient soak time produces inconsistent properties.
Temperature precision is critical throughout this stage. Even small deviations from the target can produce grain structures that are too coarse (reducing toughness) or insufficiently transformed (reducing hardness). Overheating or excessive soak times cause austenite grain coarsening, which degrades final toughness and strength.
Under-heating or under-soaking carries its own risks: incomplete carbide dissolution and insufficient transformation leave soft spots in the finished component. Metallurgical oversight and furnace calibration are non-negotiable for consistent results.
Cooling and Quenching
The cooling method determines the final microstructure. Slow cooling (furnace cool or air cool) allows atoms to reorganize gradually, producing softer, more ductile structures like pearlite. Rapid cooling (quenching in oil, water, or air) traps atoms in a stressed arrangement, producing hard, brittle structures like martensite.
Quenching media each carry different trade-offs:
- Water/Brine: Produces the fastest cooling and highest hardness but increases cracking risk due to severe thermal gradients
- Oil: Slower cooling than water, balances hardness with reduced distortion
- Air: Gentlest cooling, typically used for normalized steels or highly alloyed air-hardening steels
The choice depends on material composition and desired outcome. Cooling rate isn't always about speed—some processes deliberately slow the cooling in stages or use interrupted cooling to balance hardness and toughness without cracking.
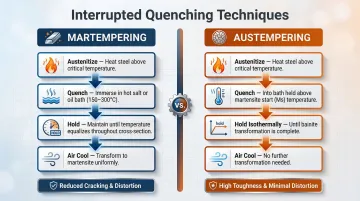
Two common interrupted quenching techniques address this balance directly:
- Martempering: The steel is quenched into a hot oil or molten salt bath (150–300°C) just above the martensite start temperature. It's held until core and surface temperatures equalize, then air-cooled. This prevents the differential expansion that causes thermal stresses and cracking.
- Austempering: Rather than cooling to martensite, the steel is held in a bath above the martensite start temperature until the austenite isothermally transforms into bainite—delivering high strength, exceptional toughness, and minimal distortion.
Main Types of Heat Treatment
Annealing
Annealing is a softening process—the metal is heated above its critical temperature and then cooled very slowly (often inside the furnace) to produce a softer, more ductile, and machinable microstructure. This process produces coarse pearlite and is commonly used before forming, drawing, or machining operations to improve workability and relieve internal stresses.
Hardening and Quenching
The metal is heated to austenitic temperature and then rapidly quenched to lock in a hard martensitic structure. This increases strength and wear resistance but typically reduces ductility. Hardening is almost always followed by tempering to reduce brittleness while retaining most of the hardness gained.
Tempering
Tempering is the correction step after hardening. The hardened metal is reheated to a lower temperature (below the critical point, typically 150-650°C) and held there to reduce internal stresses and brittleness while retaining most of the hardness. The temperature chosen determines the final hardness-toughness balance—higher tempering temperatures increase toughness but reduce hardness.
Stress Relieving and Normalizing
Stress relieving heats the metal to a sub-critical temperature (typically below 250°C to 600°C) to reduce residual stresses introduced by welding, machining, or cold forming—without altering the existing microstructure or hardness.
Normalizing heats above critical temperature (30-50°C above Ac3) and air cools to refine grain structure, eliminate effects of plastic deformation, and improve uniformity. Both processes stabilize parts before further processing.
Thermochemical Diffusion Treatments
Unlike the thermal-only processes above, this category introduces new elements into the equation: carbon, nitrogen, boron, or aluminum are diffused into the metal surface at elevated temperatures, creating an intermetallic compound layer with extreme hardness and wear resistance while the core retains toughness.
Carburizing: Carbon diffuses into austenite at 850-950°C, creating a case depth of 0.4mm to 9.0mm with surface hardness of 55-64 HRC. Low-Pressure Carburizing (LPC) at 930-1050°C eliminates intergranular oxidation and cuts cycle times, driving carbon into the steel faster and more uniformly than traditional gas carburizing.
Nitriding: Nitrogen diffuses into ferrite at 500-620°C, creating a case depth of 200-300 µm with surface hardness up to 76 HRC (900-1200 HV). The process requires no quenching, resulting in minimal distortion—ideal for precision components.
Boronizing: Boron diffuses at 700-1000°C to form FeB and Fe2B intermetallic layers with surface hardness of 1500 to 3000 HV. This provides extreme resistance to abrasive wear and galling. Boronizing is widely used for carbon steel tubing and casing in downhole drilling to prevent erosion-corrosion, creating a "saw-tooth" Fe2B layer that mechanically locks into the substrate.
Aluminizing: Aluminum diffuses to form β-NiAl or CoAl phases that oxidize in service to form a dense, self-healing α-Al2O3 (alpha-alumina) scale. This protective scale prevents high-temperature degradation and withstands extreme oxidation at temperatures up to 1100°C—critical for aerospace turbine blades and petrochemical heat exchangers.

VaporKote's boronizing and aluminizing services fall into this thermochemical diffusion category. Both processes achieve RC75+ equivalency (1500 Knoop hardness)—a hardness level that exceeds tungsten carbide cutting tools. For industries running components in high-wear or corrosive environments, that surface performance translates directly to longer service life and significantly reduced replacement and maintenance costs.
Where Heat Treatment Is Used
Heat treatment typically fits after rough machining but before final finishing—it allows the part to be shaped first, then given its final mechanical properties, with only minor post-treatment grinding or finishing required.
Critical Industrial Environments
Heat treatment is operationally critical in environments where untreated or improperly treated parts fail prematurely, driving maintenance costs and downtime:
- Oil & Gas: Downhole drilling components, mud pumps, and API 6A/6D valve seats operate in severe slurry erosion and H2S corrosion environments. Diffusion treatments like boronizing create ultra-hard surfaces that prevent erosion-corrosion failures.
- Aerospace: Landing gear made from 300M and 4340 steels requires precise austenitizing, oil quenching, and double tempering to reach 52+ HRC while preserving fracture toughness. Turbine blades in the hot-gas path rely on aluminide coatings to survive exhaust temperatures above 1100°C.
- Mining: Crusher wear liners and harvester blades need treatments that balance impact toughness with abrasion resistance—getting this wrong means brittle fracture in the field.
- Petrochemical: Reactor internals and heat exchanger tubes require strict PWHT and stress relieving. Skipping or rushing these steps leaves residual stresses that trigger stress corrosion cracking and high-temperature hydrogen attack.

The right heat treatment depends on both the base metal and the operating environment. Steel components are typically hardened and tempered; aluminum alloys are age-hardened; and parts exposed to high-temperature corrosion often require thermochemical surface treatments like aluminizing.
When standard heat treatment alone isn't enough, diffusion coating processes—boronizing and aluminizing—extend protection further. VaporKote serves these sectors from its Anaheim, CA facility, processing components up to 68 inches in diameter for both processes.
Frequently Asked Questions
What does heat treatment do to metal?
Heat treatment changes a metal's internal crystal structure (microstructure), which directly alters its mechanical properties—including hardness, ductility, toughness, and wear resistance—without changing the shape of the part.
What are the main types of heat treatment?
The primary categories include:
- Annealing, hardening/quenching, tempering, and stress relieving
- Normalizing
- Thermochemical diffusion treatments: carburizing, nitriding, boronizing, aluminizing
Each achieves a different combination of properties tailored to the application.
What is the difference between quenching, annealing, and tempering?
Quenching rapidly cools metal to maximize hardness by forming martensite. Annealing slowly cools it to maximize softness and ductility. Tempering reheats a quenched part to reduce brittleness while retaining most of its hardness.
How long do you heat treat?
Soak time depends on part thickness, material type, and the target process. A common guideline is approximately one hour per inch of thickness at temperature, though complex alloys and precision applications may require longer controlled cycles.
What metals cannot be heat treated?
Commercially pure metals like copper, 1000-series aluminum, and austenitic stainless steels (304/316) cannot be hardened by traditional heat treatment because they lack the required allotropic phase transformations. However, they can still be annealed or work-hardened.
What are heat treatments used for?
Heat treatment adjusts mechanical properties to match specific demands: increasing hardness and wear resistance, improving machinability, reducing brittleness, or stabilizing dimensions. It's used across aerospace, automotive, oil and gas, mining, and industrial manufacturing to extend component life and prevent failures.


