
Introduction
Raw aluminum has a problem: it looks capable on paper but falls short in practice. Exposed to aggressive chemicals, extreme temperatures, or constant abrasion, untreated aluminum degrades faster than most industrial operations can tolerate. A surface finish isn't optional — it's what makes the material viable.
The right surface finish extends service life, improves wear and corrosion resistance, meets regulatory requirements, and cuts long-term maintenance costs. According to the NACE IMPACT study, global corrosion costs reach $2.5 trillion annually. Between 15% and 35% of those costs are avoidable with proper corrosion control.
Choosing the right finish starts with understanding your options. This guide covers 9 of the most common aluminum surface finishes — how each works, where it performs best, and how to select the right one for your application.
TLDR
- Aluminum surface finishes improve durability, corrosion resistance, appearance, and functional performance beyond raw material capabilities
- The 9 major types span decorative options (brushed, polished) and industrial-grade coatings (hard coat anodizing, aluminizing)
- The right finish depends on your operating environment, mechanical demands, and budget — no single option works for every application
- In extreme heat or corrosive environments, diffusion-based coatings like aluminizing outperform conventional finishes
What Is Aluminum Surface Finishing?
Aluminum surface finishing is any process—mechanical, chemical, electrochemical, or thermal—applied to an aluminum part's surface to alter its properties beyond its raw state.
This covers both aesthetic treatments and functional protective coatings.
Finishing typically occurs post-machining or post-extrusion, before assembly or deployment. The base alloy matters here: high-copper alloys (2xxx series) and high-silicon cast alloys anodize poorly because intermetallic phases dissolve or cause micro-galvanic pitting during electrochemical processing.
Choosing a finish is a practical engineering decision that directly affects component lifespan. Conditions that drive the choice include:
- UV and weathering exposure
- Chemical or fluid contact
- Abrasion and wear loads
- High-temperature oxidation
Why Aluminum Surface Finishes Matter in Industrial Applications
Surface finishing delivers real operational outcomes:
- Improved corrosion resistance reduces replacement cycles
- Enhanced hardness lowers wear rates on moving or contact parts
- Correct surface prep ensures coatings bond properly for long-term adhesion
Without proper finishing, untreated aluminum oxidizes quickly in harsh environments, loses dimensional tolerance due to surface degradation, and can fail under mechanical stress ahead of schedule — a known failure point in aerospace, oil & gas, and petrochemical environments.
Finish selection is also driven by compliance requirements. Aerospace, defense, and oil refining industries mandate finishes that conform to specific ASTM, ASME, or SAE standards, which means specification alignment needs to happen before a finish is ever chosen.
9 Types of Aluminum Surface Finishes
Aluminum surface finishing is not one-size-fits-all. Different processes exist to address different functional demands, environmental conditions, and aesthetic requirements. Knowing the distinctions upfront prevents costly mismatches between finish and application.
Mill Finish
Mill finish is the natural, unprocessed surface of aluminum as it comes directly from the rolling mill or extrusion press—no secondary treatment is applied. It has a characteristic slightly dull, silvery appearance with visible tool or die marks.
Best suited for:
- Structural or internal components that will undergo further processing (welding, forming, additional coating)
- Components where surface appearance is not a priority
- Construction framing, industrial fabrication, and parts that will be painted or coated later
Key limitation: Mill finish offers no additional corrosion protection beyond aluminum's natural oxide layer. It is unsuitable for direct exposure to aggressive environments, moisture-heavy settings, or chemical contact without a secondary finish.
Anodizing (Type II — Standard)
Standard anodizing (Type II) is an electrochemical process that thickens aluminum's natural oxide layer by submerging parts in an electrolytic bath and passing an electrical current through them. The resulting anodic layer is porous enough to accept color dyes and is then sealed for durability.
Type II anodizing produces a layer typically between 0.0002" and 0.001" thick, offering solid corrosion resistance, improved surface hardness, and excellent color retention. Properly sealed Type II coatings must withstand 336 hours of neutral salt spray (ASTM B117) with a maximum of 15 isolated pits across five test specimens.
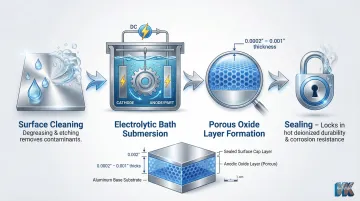
Common applications:
- Consumer electronics
- Architectural components
- Signage
- Cookware
- Medical device housings
Limitations: Type II anodizing is not suited for heavily abrasive or high-load environments where thicker, harder surface protection is required. The anodic layer can crack under significant mechanical stress, and some aluminum alloys (notably high-copper or high-silicon alloys) anodize poorly.
Hard Coat Anodizing (Type III)
Hard coat anodizing (Type III) is an advanced form of anodizing that uses lower temperatures and higher current densities to produce an oxide layer significantly thicker and denser than Type II—typically ranging from 0.001" to 0.003" or more. This results in exceptional surface hardness and wear resistance.
Type III coatings achieve hardness levels of 350–450 Vickers (60-70 Rockwell C), making them harder than many tool steels. This makes Type III ideal for high-performance industrial and military applications:
- Hydraulic components
- Aerospace parts
- Firearm components
- Sliding surfaces
- Any aluminum part subject to heavy abrasion, friction, or chemical exposure
Trade-offs:
- More expensive than standard anodizing
- Can alter dimensional tolerances (important for tight-fit machined parts)
- May reduce fatigue strength by 50–62% for cyclically loaded components
- Limits color options due to the layer's density—typically producing dark gray or black finishes
Powder Coating
Powder coating is a dry finishing process where electrostatically charged polymer powder is sprayed onto aluminum and then cured in an oven, forming a tough, continuous film. Unlike liquid paint, powder coating does not require a solvent, making it more environmentally favorable.
Standard industrial thickness ranges from 2.0 to 5.0 mils (50–125 µm). Powder coating delivers UV-resistant color finishes with strong impact and chip resistance—making it a go-to for visible, long-lasting exterior surfaces.
Common applications:
- Architectural cladding
- Outdoor furniture
- Automotive trim
- Signage
- Consumer goods where long-lasting color and moderate protection are priorities
Limitations:
- Adds measurable thickness (2–6 mils), which can affect tight dimensional tolerances
- Provides less corrosion resistance than anodizing in highly aggressive environments
- Adhesion can fail over time if the aluminum surface is not properly pre-treated (e.g., chromate or phosphate conversion coating applied first)
Studies show that combining pre-anodizing with powder coating reduces corrosion rates by nearly 1000 times compared to untreated aluminum.

Brushed Finish (Mechanical Finishing)
Brushed aluminum is a mechanically applied finish created by running abrasive belts or wire brushes across the aluminum surface in a consistent direction, producing a uniform pattern of fine parallel lines. Typical abrasive grit ranges from 180–240 for a balanced industrial satin texture, up to 320–400 for fine satin.
This reduces reflectivity while retaining a metallic appearance.
Common applications:
- Kitchen appliances
- Consumer electronics
- Interior architectural panels
- Automotive trim
- Retail display fixtures
Limitations: Brushed aluminum offers no enhanced corrosion or wear protection on its own—it is a surface texture, not a protective coating. It is typically followed by a clear anodize or lacquer coat to preserve appearance, and it is not appropriate for applications demanding dimensional precision or mechanical durability.
Polished Aluminum
Polished aluminum is a finish achieved through progressive mechanical buffing with increasingly fine abrasives until the surface reaches a mirror-like, highly reflective appearance. It can also be enhanced chemically through bright dip processes (hot phosphoric-nitric acid baths at 190–220°F) prior to anodizing.
Bare, polished aluminum provides excellent reflectivity (>91% from 200–2000nm).
Best use cases:
- Lighting reflectors
- Architectural accents
- Marine fittings
- High-end consumer products
- Solar energy concentrators where reflectivity serves a functional purpose
Trade-offs: Polished surfaces are highly susceptible to fingerprints, scratches, and oxidation without a protective clear coat or anodize seal. The buffing process is labor-intensive and costly, and polished finishes are impractical for functional industrial components exposed to abrasion or harsh environments.
Chemical Conversion Coating (Alodine / Chromate)
Chemical conversion coating (commonly known as Alodine or chromate conversion coating) is a chemical treatment that reacts with the aluminum surface to form a thin, electrically conductive protective layer—typically chromate-based (hexavalent or trivalent)—without significantly affecting dimensional tolerances.
Primary strengths:
- Combines corrosion protection with electrical conductivity — anodizing cannot offer both simultaneously
- Indispensable in aerospace, defense, and electronics applications where grounding or electromagnetic shielding is required
- Used as a primer adhesion layer before painting or powder coating
Chemical conversion coatings are governed by MIL-DTL-5541F:
- Class 1A: Provides maximum protection against corrosion for painted or unpainted items
- Class 3: For electrical applications requiring low electrical resistance (<5,000 microhms per square inch)
Important regulatory note: Hexavalent chromate coatings face increasing environmental and regulatory restrictions (RoHS, REACH directives). Trivalent chromate alternatives are available but may offer slightly less corrosion performance. Because the coating is thin and soft relative to anodizing, it is not suited for surfaces exposed to abrasion or mechanical wear — pair it with a topcoat when durability is also required.
Bead Blasting / Sandblasting
Bead blasting is a mechanical process that propels small spherical media (glass beads, aluminum oxide, or steel shot) at the aluminum surface under pressure to create a uniform matte or satin texture by removing surface irregularities, tool marks, and oxidation.
Uses:
- Standalone finish for uniform visual texture (common in aerospace interiors, consumer electronics, sporting goods)
- Surface preparation step before anodizing, painting, or powder coating to improve adhesion
Critical warning: MIL-A-8625F explicitly prohibits the use of iron-containing abrasives (like steel wool or steel wire) prior to anodizing, as ferrous particles embed in the aluminum substrate and accelerate galvanic corrosion.
Limitations: Bead blasting alone offers no corrosion or wear protection and can embed contaminants into the surface if not properly controlled. Media selection and blast pressure must be carefully matched to the aluminum alloy to avoid surface stress, deformation, or excessive material removal.
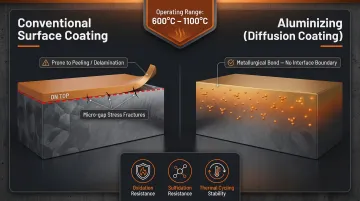
Aluminizing (Diffusion Coating)
Aluminizing is a thermochemical diffusion process in which aluminum is deposited into the surface of a base metal (steel, nickel alloys, or other substrates) through chemical vapor deposition, forming an intermetallic compound that becomes an integral part of the base material—not simply a surface layer sitting on top.
This is fundamentally different from conventional aluminum surface finishes, as the aluminum becomes metallurgically bonded into the substrate.
Performance outcomes:
Aluminized components achieve outstanding resistance to high-temperature oxidation, sulfidation, and corrosion—making this process essential for components in:
- Petrochemical reactors
- Oil refining equipment
- Heat exchangers
- Aerospace turbine parts
- High-temperature industrial furnace hardware
These intermetallics form a protective alumina scale that provides exceptional protection at extreme temperatures (600°C to 1100°C).
Key differentiator: Because the coating is a diffusion-bonded intermetallic layer rather than a surface film, it cannot peel, chip, or delaminate—and it maintains protection even under thermal cycling and mechanical stress that would destroy paint, powder coating, or even anodizing.
VaporKote's aluminizing process uses chemical vapor deposition to form this intermetallic layer on industrial components. VaporKoted parts consistently outperform conventional surface treatments in wear and corrosion testing. The company processes components up to 68" in diameter and works to ASTM, ASME, SAE, and API engineering codes.
How to Choose the Right Aluminum Surface Finish
Application requirements determine the right finish. The same aluminum part used in an architectural panel versus a petrochemical heat exchanger can require completely different finishing approaches — and what works in one context can fail quickly in another.
Operational Environment
Assess whether the part will face moisture, UV exposure, chemical contact, abrasion, high temperatures, or a combination:
- Type III hard coat suits high-wear mechanical contact
- Aluminizing suits high-temperature oxidizing environments
- Powder coating suits outdoor architectural applications with UV exposure
- Chemical conversion coating suits electronics requiring electrical conductivity
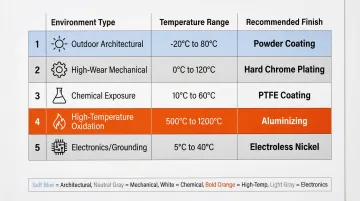
| Environment Type | Temperature Range | Recommended Finish |
|---|---|---|
| Outdoor architectural | Ambient | Powder coating (AAMA 2605) |
| High-wear mechanical | Ambient to 200°F | Type III hard coat anodizing |
| Chemical exposure | Ambient to 400°F | Type II anodizing or chem-film |
| High-temperature oxidation | 600°C to 1100°C | Aluminizing (diffusion coating) |
| Electronics/grounding | Ambient | Chemical conversion (Class 3) |
Dimensional Tolerance and Post-Finishing Fit
Some finishes add measurable thickness that can affect how machined parts fit into assemblies:
- Type III anodizing: Increases part dimensions by one-half of the applied coating thickness per surface (a 0.004" coating, for example, requires a 0.002" pre-machining allowance per surface)
- Powder coating: Adds 2–6 mils of thickness
- Chemical conversion coating and mill finish: Add negligible thickness
Tight-tolerance CNC parts require finish selection at the design stage. ASME Y14.5 requires engineering drawings to explicitly state whether dimensions apply before or after coating.
Aesthetic vs. Functional Priority
Decorative applications (signage, consumer products, architectural) can favor:
- Brushed finishes
- Polished finishes
- Powder-coated finishes
Structural and industrial components should prioritize protection and durability metrics over appearance:
- Type II or Type III anodizing
- Chemical conversion coating
- Aluminizing
Understanding these priorities also helps avoid the most common finish selection errors — especially when engineers default to familiar processes without evaluating fit.
Common Selection Mistakes to Avoid
- Over-specifying a finish when a simpler option meets all requirements — adds cost without benefit
- Underestimating long-term maintenance: a finish that looks good initially but degrades in service costs more over time
- Defaulting to a familiar process without checking whether it suits the specific alloy or operating conditions
- Skipping pre-treatment steps — for example, conversion coating before powder coating — that the finish needs to bond properly
- Applying Type III hardcoat to cyclically loaded components without accounting for the 50%+ reduction in fatigue strength
Conclusion
Aluminum surface finishes span a wide spectrum—from simple mill finish and decorative brushed surfaces to high-performance anodizing, chemical conversion coatings, and diffusion-based aluminizing. Each serves a distinct functional or aesthetic purpose.
Choosing the wrong finish leads to premature failure, increased maintenance costs, and unplanned downtime. Match your selection to:
- Operating environment — temperature range, chemical exposure, humidity
- Mechanical demands — wear loads, hardness requirements, fatigue stress
- Regulatory requirements — industry standards such as ASTM, ASME, or API
- Long-term cost of ownership — upfront application cost vs. service life extension
For components in high-temperature or corrosive industrial environments, diffusion-based finishes like aluminizing offer protection that decorative or electrochemical processes simply can't match. VaporKote has specialized in aluminizing and thermal diffusion coatings since 1987 — reach out at sales@vaporkote.com or (714) 632-8607 if you need guidance on the right finish for your application.
Frequently Asked Questions
What is an aluminum surface finish?
An aluminum surface finish is any treatment—mechanical, chemical, electrochemical, or thermal—applied to aluminum to improve its appearance, durability, corrosion resistance, or functional performance beyond the natural state of the raw metal.
What is the best finish for aluminum?
There is no single "best" finish — the right choice depends entirely on the application. Anodizing suits general industrial use, powder coating excels for outdoor color durability, and aluminizing is the preferred option for high-temperature or chemically aggressive environments.
What is the difference between anodizing and powder coating for aluminum?
Anodizing is an electrochemical process that converts the aluminum surface itself into a protective oxide layer, while powder coating applies an external polymer film via electrostatic spray and heat curing. Anodizing offers better corrosion resistance and hardness; powder coating excels in color variety and UV resistance.
Which aluminum surface finish is best for high-temperature or corrosive industrial environments?
Conventional finishes like anodizing or powder coating are not designed for extreme heat. For petrochemical, oil refining, and high-temperature industrial applications, aluminizing (diffusion coating) is the preferred solution as it creates a metallurgically bonded intermetallic layer that resists oxidation and sulfidation at elevated temperatures.
Is mill finish aluminum suitable for use without any additional treatment?
Mill finish works for structural or internal components that will be further processed or shielded from the environment. For parts exposed directly to moisture, chemicals, or outdoor conditions, a secondary finish is strongly recommended — mill finish alone provides minimal corrosion protection.
Does the aluminum alloy affect which surface finish can be applied?
Yes. Alloy composition significantly impacts finish compatibility. High-silicon or high-copper alloys (such as 2xxx or cast 356 series) anodize differently than high-purity alloys, and some alloys respond poorly to certain chemical treatments. Always verify finish compatibility with the specific alloy before selecting a finishing process.


