
Introduction
Premature component failure due to wear, friction, and corrosion costs manufacturers millions in unplanned downtime and replacement parts. In high-stress environments—aerospace turbines, automotive powertrains, petrochemical processing equipment—surface degradation is inevitable.
Engineers rely on advanced surface coatings to extend service life and cut total cost of ownership. Physical Vapor Deposition (PVD) is one of the most effective solutions available, delivering ultra-hard, wear-resistant coatings that bond at the atomic level.
This article explains what PVD coating is, how the deposition process works step by step, and the different methods used in industrial applications. It also covers measurable performance benefits, the industries that rely on PVD most, and how it compares to diffusion-based processes that can deliver even greater hardness in the most demanding applications.
TLDR:
- PVD vaporizes solid materials and deposits ultra-thin, high-performance films bonded atom-by-atom to the substrate
- Vaporization, vacuum transport, optional reactive gas, and condensation combine to form dense, adhesive coatings
- Sputtering, cathodic arc, and evaporation each suit different geometries and performance needs
- PVD coatings deliver hardness up to 3,500 HV, friction coefficients as low as 0.1, and eliminate hazardous chemicals used in chrome plating
- Industries from aerospace to medical devices rely on PVD for cutting tools, engine components, implants, and decorative hardware
What Is PVD Coating?
Physical Vapor Deposition (PVD) is a vacuum-based process where solid source materials are converted into vapor and deposited onto a substrate as an ultra-thin, high-performance film. Unlike surface treatments that simply adhere to a component, PVD creates coatings through atom-by-atom deposition, where vaporized material travels through a vacuum or low-pressure environment and condenses directly onto the substrate surface. The result is a metallurgically integrated layer — not a superficial coating applied on top.
The resulting film is typically a metal or metal-ceramic compound — titanium nitride (TiN), chromium nitride (CrN), aluminum titanium nitride (AlTiN), or diamond-like carbon (DLC). Because the coating bonds at the atomic level, it delivers adhesion strength, surface hardness, and finish quality that conventional treatments can't replicate.
Core Components of a PVD System:
A PVD system consists of several essential elements working together under controlled vacuum conditions:
- A sealed vacuum chamber that maintains the low-pressure environment required for clean vapor transport
- A high-purity source material (the "target") — metal, alloy, or ceramic — that vaporizes to form the coating
- A substrate holder that positions and rotates components for uniform coverage
- An energy source (heat, plasma, or electron beam) that drives vaporization of the target material
These components determine the final coating characteristics, including thickness. Decorative thin films typically fall below 0.5 microns, while functional hardcoatings run 2 to 5 microns. Going beyond 5 microns rarely improves performance and can introduce residual stress into the part.
How Does the PVD Process Work?
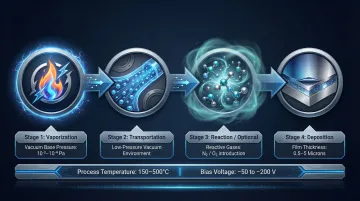
The PVD process consists of four fundamental stages operating under precise vacuum control:
1. Vaporization
The coating material — called the target — converts from solid to vapor using physical energy. Four methods achieve this:
- Heat – Thermal evaporation raises the target temperature until atoms escape the surface
- Plasma bombardment – High-energy ions strike the target, dislodging atoms (sputtering)
- Electron beam – A focused beam melts and vaporizes the source material
- Electric arc – A high-current arc creates localized vaporization (cathodic arc)
The vaporization occurs inside a vacuum chamber evacuated to base pressures as low as 10⁻³ to 10⁻⁴ Pa, ensuring minimal contamination.
2. Transportation
Vaporized atoms, ions, or molecules travel through the low-pressure vacuum environment toward the substrate. Under vacuum, the mean free path of atoms increases dramatically — allowing direct, uninterrupted travel from target to substrate without collisions that would scatter the vapor or introduce atmospheric contaminants.
3. Reaction (Optional)
In reactive PVD, gases such as nitrogen or oxygen are introduced into the chamber during deposition. These reactive gases combine with the vaporized metal atoms in transit, forming compound coatings:
- Nitrogen + Titanium → Titanium Nitride (TiN)
- Nitrogen + Chromium → Chromium Nitride (CrN)
- Oxygen + Aluminum → Aluminum Oxide (Al₂O₃)
- Nitrogen + Titanium + Aluminum → Titanium Aluminum Nitride (TiAlN)
Reactive coatings typically reach hardness values of 2,000–3,000 HV — well beyond what pure metal films achieve.
4. Deposition, Film Formation, and Process Control
Vapor atoms condense onto the substrate surface, nucleating and growing into a dense, tightly bonded thin film. Individual atoms find energetically favorable positions on the substrate and bond to neighboring atoms at the atomic scale. The result is a coating typically 0.5 to 5 microns thick — thin enough to preserve tight tolerances, yet hard enough to outperform many bulk materials.
Achieving target film properties requires precise control of multiple parameters throughout the deposition cycle:
- Chamber pressure – Magnetron sputtering operates at 0.1–1.5 Pa, while cathodic arc systems maintain 10⁻³ to 10⁻⁴ Pa base pressure
- Substrate temperature – Typically 150°C to 500°C, below the tempering temperature of most steels to preserve core material properties
- Deposition rate – Usually up to 10 nm/s, controlled to balance film quality and production throughput
- Substrate bias voltage – Typically -50 V to -200 V, accelerating positive ions toward the substrate to enhance adhesion, density, and deposition rate
Process engineers monitor and adjust these parameters in real time to hit target thickness, hardness, residual stress, and adhesion strength.
Types of PVD Deposition Methods
Sputtering
Sputtering uses a plasma—typically argon ions—to bombard a target material. Energetic ions strike the target surface, ejecting atoms that travel to and coat the substrate. This method offers several advantages:
- Sputtered atoms arrive from multiple angles, improving coverage on sidewalls and complex geometries
- The bombardment process creates tightly packed atomic structures with high film density
- Works across a wide range of materials, including alloys and compounds that are difficult to evaporate
Magnetron sputtering, the most common industrial variant, uses magnetic fields to confine the plasma near the target, increasing ionization efficiency and deposition rates.
Evaporation (Thermal and Electron Beam)
Evaporation methods heat a source material until it vaporizes:
- Thermal evaporation – Resistive heating or induction raises the source temperature until atoms escape
- Electron beam evaporation – A focused electron beam melts and vaporizes the target with precise energy control
Evaporation is highly directional, making it well-suited for lift-off photolithography applications and deposition of thicker films. However, the line-of-sight nature limits conformal coverage on complex three-dimensional geometries compared to sputtering.
Cathodic Arc Deposition
Cathodic arc employs a low-voltage, high-current electric arc to vaporize material from a solid cathode target. This method produces near 100% ionization with high kinetic energy (20–200 eV), promoting highly dense, adherent film growth. The exceptional adhesion makes cathodic arc the preferred method for hard coatings like TiN on cutting tools and wear-resistant industrial components.
The primary limitation is macrodroplet formation—small particles ejected from the cathode that can degrade film quality. Filtered Cathodic Arc (FCA) technology uses magnetic fields to separate the plasma from neutral atoms and macroparticles, delivering the adhesion benefits while maintaining smooth surface finish.

High Power Impulse Magnetron Sputtering (HiPIMS)
HiPIMS applies very short, high-power pulses to dramatically increase ionization of sputtered material—achieving film density and adhesion comparable to cathodic arc, while retaining the droplet-free, smooth surface finish that conventional sputtering is known for. The result is a process well-suited to precision components where both mechanical performance and surface quality are non-negotiable.
Pulsed Laser Deposition (PLD)
PLD uses a high-powered laser to ablate the target material, creating a vapor plume that deposits onto the substrate. It is used mainly in research settings where exact stoichiometric control of complex films is critical—such as high-temperature superconductors or ferroelectric oxides—rather than high-volume industrial production.
Key Benefits of PVD Coatings
Exceptional Hardness and Wear Resistance
PVD coatings dramatically increase surface hardness, cutting wear and extending component service life. Common industrial PVD coatings achieve hardness levels that far exceed uncoated metals and traditional plating:
- TiN (Titanium Nitride): 2,000–2,800 HV
- CrN (Chromium Nitride): ~1,750 HV
- AlTiN/TiAlN: 3,000–3,500 HV
- DLC (Diamond-Like Carbon): 1,500–2,000+ HV
For context, hard chrome plating typically achieves 800–1,200 HV. The superior hardness of PVD coatings translates directly to longer service intervals, reduced maintenance costs, and fewer unplanned shutdowns.

Superior Corrosion and Chemical Resistance
The dense, pinhole-reduced structure of PVD films creates a highly effective barrier against corrosion, oxidation, and chemical attack. The atom-by-atom deposition process produces coatings with minimal defects, blocking corrosive media from the substrate. This makes PVD coatings particularly valuable in aggressive environments — petrochemical processing, marine applications, and chemical handling equipment.
Low Friction Coefficient
WC/C and DLC coatings deliver some of the lowest friction coefficients available in surface engineering. Uncoated steel exhibits friction coefficients of 0.74–0.80 under dry conditions, while WC/C and DLC coatings achieve 0.1–0.2—a reduction of more than 75%. This friction reduction benefits engine parts, bearings, cutting tools, and any application where sliding contact occurs.
Aesthetic Versatility
PVD coatings can be applied in a wide spectrum of colors and finishes—gold, black, titanium blue, bronze, rose gold—without sacrificing durability. Unlike paint or anodizing, PVD decorative finishes hold their hardness and scratch resistance over time — making them a practical choice for watches, architectural hardware, and automotive trim where appearance and longevity both matter.
Environmental and Process Advantages
PVD is a relatively clean, dry process with minimal use of hazardous chemicals compared to electroplating or chemical treatments. The vacuum-based process uses only solid metals and inert or reactive gases (nitrogen, argon, oxygen), producing no toxic emissions or hazardous wastewater sludges.
Hard chrome electroplating, by contrast, relies on chromic acid baths containing hexavalent chromium (Cr(VI)), a known human carcinogen strictly regulated by OSHA and the EPA. PVD CrN coatings eliminate Cr(VI) exposure entirely while delivering superior hardness and performance.
Industrial Applications of PVD Coatings
The global PVD market was valued at $2.99 billion in 2023 and is projected to reach $4.99 billion by 2030, driven by demand from tooling, aerospace, medical, and electronics sectors.
Cutting Tools and Tooling
PVD coatings like TiN, Ti(C,N), and AlTiN are widely applied to drill bits, end mills, carbide inserts, and molds to reduce wear, allow higher cutting speeds, and extend tool life. This represents one of the largest and most established industrial uses of PVD. Studies on high-speed machining of Inconel 718 using TiAlN-coated carbide tools achieved tool life up to 95.38 minutes, while multi-layered TiCN coatings increased tool life by an average of 50% in customer tests. The thermal barrier and oxidation resistance provided by these coatings enable dry machining and higher material removal rates.
Aerospace, Automotive, and Energy
PVD-coated components benefit from improved wear resistance and thermal stability in high-stress, high-temperature operating conditions. Applications include:
- Turbine blades – Thermal barrier coatings and erosion protection
- Engine valves and valve train components – DLC coatings reduce friction and prevent scuffing during starved-lubrication events
- Fuel system parts – Corrosion resistance in aggressive fuel environments
- Piston rings – WC/C coatings reduce friction and wear in combustion engines
In aggregate, these performance gains translate to longer service intervals and measurable reductions in fuel consumption — a priority for aerospace OEMs and automotive manufacturers under tightening emissions standards.

Medical Devices, Electronics, and Decorative
PVD serves specialized roles across three additional sectors:
- Medical devices – TiN and DLC coatings certified biocompatible under ISO 10993-1 are used on orthopedic implants and surgical instruments, reducing wear debris and ion release while promoting bone integration
- Electronics and semiconductors – PVD TiN and TaN form diffusion barrier layers (often ~1 nm thick) in copper interconnects, preventing copper migration into silicon substrates during integrated circuit fabrication
- Industrial hardware and fixtures – PVD delivers durable, scratch-resistant finishes on architectural hardware, industrial fixtures, and precision instruments, combining corrosion resistance with a consistent surface appearance
PVD vs. Other Coating Methods
PVD vs. CVD (Chemical Vapor Deposition)
PVD and CVD represent fundamentally different approaches to thin film deposition:
| Factor | PVD | CVD |
|---|---|---|
| Process Temperature | 150°C–500°C — below tempering range of most steels, no post-treatment needed | 800°C–1,050°C — often requires post-coating heat treatment for steel substrates |
| Residual Stress | Compressive stress (especially cathodic arc) — inhibits surface crack initiation | Tensile stress from thermal mismatch during cooldown — can cause micro-cracking |
| Conformality | Line-of-sight process — complex internal geometries require part rotation or specialized fixturing | Gas-phase precursors coat complex geometries and deep internal diameters uniformly |
For the most demanding heavy-industrial applications—petrochemical processing, oil drilling, mining—diffusion-based coatings can outperform both. VaporKote's boronizing and aluminizing processes achieve RC75+ hardness equivalency (1,500 Knoop) and form metallurgically bonded intermetallic compounds that outlast conventional PVD in extreme erosion and corrosion environments.
PVD vs. Electroplating (Chrome and Gold)
PVD offers substantial advantages over traditional electroplating:
- Regulatory compliance: Hard chrome plating uses hexavalent chromium (Cr(VI)) baths, a known carcinogen regulated by OSHA (PEL of 5 µg/m³) and the EPA. PVD CrN coatings eliminate Cr(VI) exposure entirely while achieving higher hardness (~1,750 HV vs. 800–1,200 HV for hard chrome).
- Wear performance: PVD coatings are harder and more wear-resistant than electroplated chrome or gold. Chrome plating suffers from inherent microcracking; gold plating offers limited hardness. PVD delivers functional performance and aesthetic versatility in one step.
- Process cleanliness: Electroplating generates hazardous wastewater sludge (EPA F006 waste) and toxic mist. PVD is a dry, vacuum-based process with no liquid waste streams.

Choosing the Right Coating
Selecting the optimal coating technology depends on several key factors:
- Substrate material – Temperature sensitivity, surface hardness, and compatibility
- Operating temperature – Maximum service temperature and thermal cycling
- Geometry complexity – Internal features, aspect ratios, and accessibility
- Required hardness and corrosion resistance – Performance specifications and environment severity
- Batch size and cost – Production volume and total cost of ownership
PVD excels for temperature-sensitive substrates requiring hard, wear-resistant coatings with excellent adhesion. CVD is preferred for complex geometries and applications requiring conformal coverage at high temperatures. For extreme wear and corrosion in heavy industrial environments, diffusion-based processes may offer superior longevity and cost-effectiveness.
Frequently Asked Questions
What is a PVD coating?
PVD coating is a vacuum-based deposition process that converts solid source materials into vapor, which then condenses onto a substrate as a thin metal or metal-ceramic film — typically 2–5 microns thick. The atom-by-atom bonding improves hardness, wear resistance, corrosion protection, and appearance.
What are the steps of PVD process?
PVD follows four sequential steps:
- Vaporization — physical energy converts solid source material into vapor inside a vacuum chamber
- Transportation — vapor atoms travel to the substrate through a low-pressure environment
- Reaction (optional) — reactive gases are introduced to form compound coatings
- Deposition — vapor condenses and bonds tightly as a thin film onto the surface
What are the different types of PVD deposition?
The five primary PVD methods are sputtering, thermal evaporation, electron beam evaporation, cathodic arc deposition, and pulsed laser deposition. Sputtering and cathodic arc are most common in industrial hard-coating applications.
What are the pros and cons of PVD?
PVD delivers exceptional hardness (up to 3,500+ HV), superior wear and corrosion resistance, low friction, and cleaner processing than electroplating. The main limitations are line-of-sight constraints on complex internal geometries, higher equipment costs, and film thicknesses (2–5 microns) that may fall short for applications requiring substantial material build-up.
Is PVD coating better than chrome?
PVD generally outperforms chrome plating in hardness (1,750–3,500 HV vs. 800–1,200 HV), scratch resistance, and environmental safety (no hexavalent chromium). Chrome remains an option for thick build-up or tight-budget situations, but PVD CrN coatings eliminate hexavalent chromium concerns while delivering superior performance.
Can PVD be cured?
PVD coatings do not require a separate curing step like paint or powder coatings. The film forms and bonds to the substrate during the deposition process itself inside the vacuum chamber. Parts are ready for use once the process cycle is complete and the chamber is vented to atmospheric pressure.


