Introduction
Metal components fail from the surface first. In manufacturing, oil & gas, mining, aerospace, and petrochemical operations, equipment doesn't typically break down due to bulk material weakness—it breaks down through surface wear and corrosion. Global corrosion costs reach approximately $2.5 trillion annually, equivalent to 3.4% of global GDP, with the majority of this economic burden stemming from preventable surface degradation.
Thin film deposition for metals is a process in which a very thin layer of material is deposited onto a metal substrate—through physical or chemical means—to alter its surface properties without significantly changing its dimensions. For engineers, maintenance managers, and procurement leads, it's a direct way to extend component life and reduce the unplanned downtime that drives maintenance costs up.
This guide covers how thin film deposition works, the key methods used on metals (PVD, CVD, and IVD), what drives coating performance, and when the process fits your application—and when it doesn't.
TLDR
- Thin film deposition applies ultra-thin protective layers to metal surfaces, improving hardness, corrosion resistance, and wear performance without altering dimensional tolerances
- PVD delivers extreme hardness (up to 3300 HK) in coatings as thin as 0.5–5 µm, but requires line-of-sight access to all surfaces
- CVD diffusion coatings bond metallurgically within the substrate, covering complex geometries uniformly at RC75+ hardness
- IVD aluminum is the aerospace-standard cadmium replacement — rated to 500°C and free of hydrogen embrittlement risk
- Choosing the wrong deposition method for your substrate, geometry, or operating environment results in premature failure and unnecessary cost
What Is Thin Film Deposition for Metals?
Thin film deposition is a surface engineering process that adds a layer — anywhere from nanometers to tens of microns thick — onto a metal substrate to change its surface characteristics. The deposited layer modifies hardness, corrosion resistance, and friction behavior without touching the bulk material properties.
The intended outcome falls into two categories. First, the deposited layer can act as a protective barrier, blocking oxidation, corrosion, and abrasion from reaching the base metal. Second, in diffusion-based processes, the coating material reacts with the substrate to form an intermetallic compound — a new phase chemically bonded into the surface rather than simply sitting on top of it.
How thin film deposition differs from related processes:
- Thermal spray coatings apply thick, sometimes porous layers (100+ microns) that can shift part dimensions enough to require post-coating machining
- Electroplating deposits metal via chemical bath immersion, which carries hydrogen embrittlement risk for high-strength steels
- Thin film deposition operates in vapor phase — either in vacuum or controlled atmosphere — producing denser, more adherent coatings that hold dimensional tolerances typically under 50 microns
That precise control over coating thickness, composition, and microstructure is what makes these processes the go-to choice in aerospace, oil and gas, and precision manufacturing — anywhere a failed surface means a failed component.
Why Thin Film Deposition Is Used in Industrial Metal Applications
The Economic Imperative
The global cost of corrosion is estimated at $2,505 billion, representing 3.4% of global GDP. Applying available corrosion control practices, industries can recover between 15% and 35% of those costs—$375 billion to $875 billion annually. These figures exclude the indirect costs of safety incidents, environmental releases, and forced operational outages.
Industrial environments demand specific performance characteristics that thin film deposition addresses:
- Maintains dimensional stability without altering part tolerances or requiring remachining
- Bonds metallurgically to survive repeated thermal cycling and shock
- Resists attack from hydrocarbons, caustic media, and aggressive process fluids
- Achieves surface hardness that outlasts the base substrate
What Happens Without Surface Treatment
Components operating without surface treatment in abrasive or corrosive environments fail at the surface first. Erosion, pitting, and galling initiate failure long before bulk material fatigue becomes a concern. This surface-initiated degradation leads to:
- Unplanned downtime during critical production periods
- Emergency component replacement at premium costs
- Cascading failures as worn components damage mating parts
- Reduced operational efficiency as clearances open and tolerances degrade
These failure modes have also shaped how regulators and standards bodies approach surface treatment requirements.
Regulatory and Standards Requirements
Certain industries operate under strict codes that dictate acceptable surface treatments. MIL-DTL-83488D governs ion vapor deposited aluminum coatings for aerospace applications. API TR 934-F addresses hydrogen embrittlement concerns in high-pressure hydrogen service. ASTM B117 establishes salt spray testing protocols for corrosion resistance validation.
In oil & gas, aerospace, and defense, many specifications formally require thin film deposition processes that satisfy these standards for defined component classes.
The ROI Case
The financial case for surface treatment is well-documented. U.S. Department of Defense corrosion mitigation projects document validated ROIs ranging from 8.8 to 56. Specific validated returns include:
- ROI 56: Magnesium-rich primer for chrome-free aircraft coating systems
- ROI 15: Ceramic anode upgrades for cathodic protection
- ROI 8.8: Coating systems for cathodic protection and fire resistance on metal structures
For industrial operations, coated components routinely outperform untreated materials on both life and maintenance cost. The savings show up in fewer emergency replacements, longer intervals between shutdowns, and components that don't degrade neighboring parts.
How Thin Film Deposition Works
Regardless of the specific method, thin film deposition on metals follows three fundamental stages: surface preparation of the substrate, creation and transport of the deposition material in vapor or ion form, and bonding or condensation of that material onto the metal surface under controlled conditions.
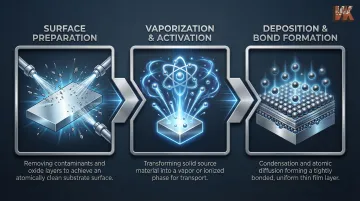
Step 1: Surface Preparation
Substrate cleanliness and surface condition are the single most critical variables in coating adhesion. Contamination from oxides, oils, or surface irregularities at the interface leads to delamination under thermal or mechanical stress.
Common preparation methods include:
- Abrasive blasting: Creates mechanical anchoring through surface roughness; can introduce residual stress
- Chemical etching: Removes oxides and activates the surface chemically; must be followed by thorough rinsing
- In-situ vacuum cleaning: Argon glow discharge (ion bombardment) removes native oxides immediately before deposition without exposing the surface to atmosphere
Each method affects the final bond differently. Ion etching in vacuum produces the cleanest, most reactive surface but requires integrated processing equipment. Chemical etching is effective for batch processing but introduces handling steps between cleaning and coating.
Step 2: Vaporization or Activation of the Coating Material
The source material—metal, alloy, or compound—is transformed into a vapor, ion, or reactive gas phase. The transformation method determines the energy of deposition and, by extension, the coating density and adhesion strength.
Two broad approaches drive this transformation:
- Physical vaporization: The source is heated until it evaporates, or bombarded by plasma that ejects atoms from a solid target (sputtering)
- Chemical decomposition: Volatile metallic compounds (metal halides or organometallics) break down at the heated substrate surface, releasing the coating element
Higher deposition energy generally produces denser coatings with better adhesion. The trade-off: excessive energy can introduce residual stress or damage temperature-sensitive substrates.
Step 3: Deposition and Bond Formation
Vaporized or ionized material travels to the substrate and either condenses (in PVD) or chemically reacts with the metal surface (in CVD/diffusion) to form the coating.
In diffusion-based processes, the coating material actually diffuses into the surface lattice, forming an intermetallic compound rather than a discrete separate layer. This results in a metallurgical bond rather than a mechanical one. The coating is not a layer sitting on top but a transformed zone of the metal itself, with significantly higher adhesion and hardness than surface-only coatings.
Thin Film Deposition Methods Used in Metal Protection
While dozens of thin film techniques exist, three are most relevant to industrial metal protection: Physical Vapor Deposition (PVD), Chemical Vapor Deposition (CVD)/diffusion coating, and Ion Vapor Deposition (IVD). Each has a distinct mechanism, performance envelope, and application fit.
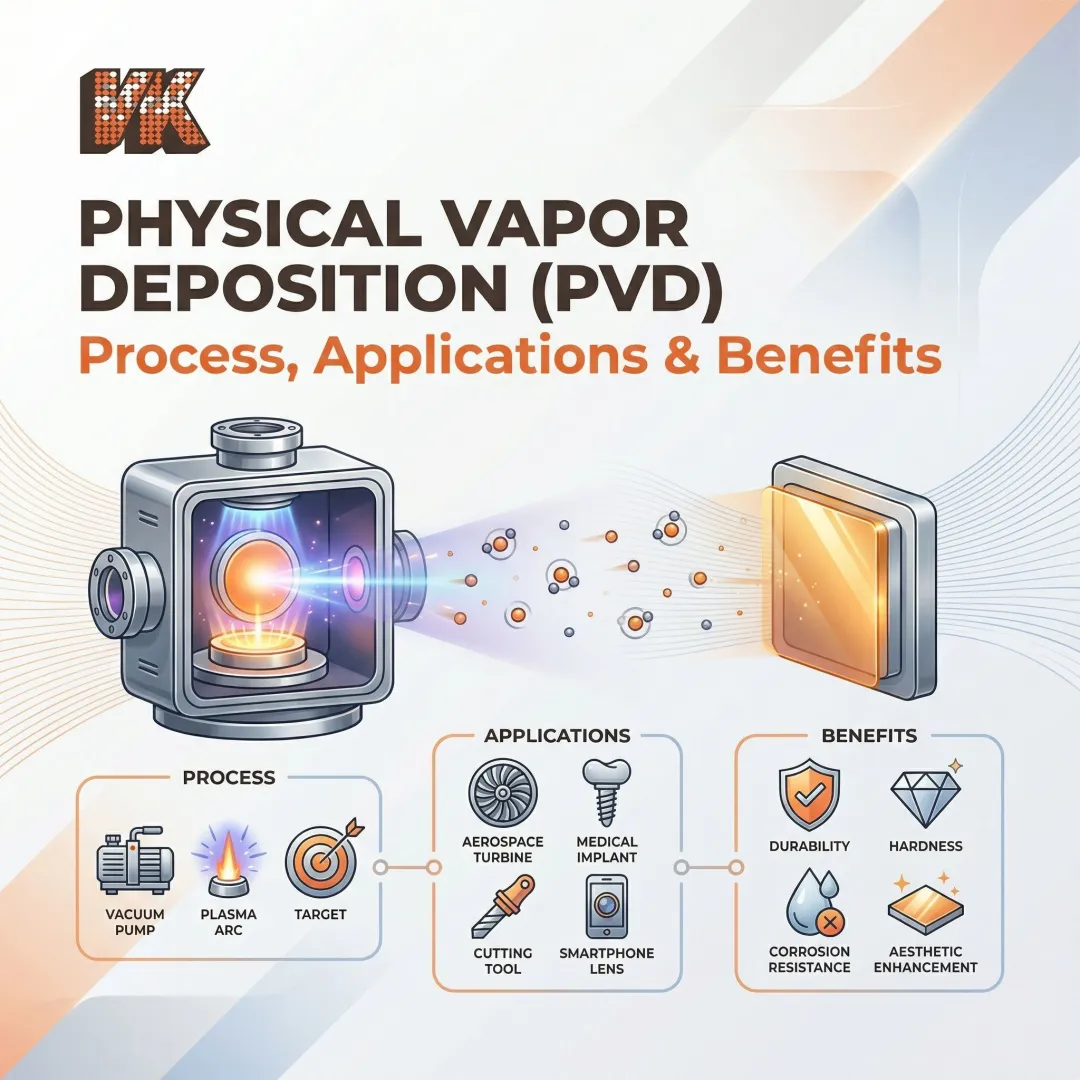
Physical Vapor Deposition (PVD)
PVD is a vacuum-based line-of-sight process where solid source material is vaporized via sputtering or evaporation and condenses on the metal substrate. Coatings are typically 0.5–5 microns thick, very hard, and used where tight dimensional tolerances and surface hardness are required.
Common PVD coating materials for industrial metals:
| Coating | Microhardness | Max Service Temp | Primary Benefit |
|---|---|---|---|
| Titanium Nitride (TiN) | ~2300 HK | 600°C | General purpose abrasion resistance; high-temperature stability |
| Chromium Nitride (CrN) | ~1750 HK | 700°C | Superior corrosion and oxidation resistance; replaces hard chrome |
| Titanium Aluminum Nitride (TiAlN) | ~3300 HK | 900°C | Extreme wear resistance and hot hardness for high-speed machining |

In turning operations on Ta-2.5W alloys, TiAlN-coated carbide tools achieved 117% longer tool life compared to uncoated tools. In tungsten heavy alloy machining, TiAlN-coated tools reached 15 km cutting distance before reaching wear failure criteria.
The line-of-sight limitation: PVD's fundamental constraint is that material transfers in a straight path from source to substrate. Areas not in direct view of the vapor flux receive little to no coating. In complex geometries with high aspect ratio holes, geometrical shadowing prevents coating growth on lower sidewalls, leading to pinholes and uneven coverage.
Industrial PVD reactors use multi-axis planetary fixturing to rotate substrates, randomizing the angle of incidence. This improves coverage on complex external shapes but cannot solve the fundamental limitation for deep recesses, internal bores, or blind holes.
Chemical Vapor Deposition (CVD) and Diffusion Coating
CVD is a process where reactive gas-phase compounds decompose or react at the heated metal surface, forming a coating that chemically bonds with the substrate. In diffusion coating specifically—aluminizing and boronizing—the coating element diffuses into the surface lattice to form a hard intermetallic compound.
Key industrial advantages:
- Covers complex parts conformally, including internal passages and recessed features — no line-of-sight required
- Chemically integrates with the substrate, eliminating delamination risk
- Boronized surfaces achieve 1650–1850 HK0.1 (FeB phase) and 1400–1650 HK0.1 (Fe2B phase), equivalent to RC75+ hardness
- Scales to large components, with industrial furnaces handling parts several feet in diameter
| Diffusion Process | Temp Range | Active Species | Resulting Phase | Primary Application |
|---|---|---|---|---|
| Boronizing | 700–1000°C | Boron (B) | FeB / Fe2B | Extreme abrasive wear protection for pumps and tubing |
| Aluminizing | 800–1100°C | Aluminum (Al) | β-NiAl / FeAl | High-temperature oxidation and hot corrosion resistance |

VaporKote, for instance, formulates powder mixes on-site and processes large-format parts using industrial-scale furnaces — an approach well-suited to the scale demands of petrochemical, oil and gas, and geothermal applications.
The process is proven in demanding environments. CVD processes are routinely used to coat 24-foot straight heat exchanger tubes to prevent fouling and corrosion in petrochemical plants, while thermal diffusion boronizing is successfully applied to large-size spools, long tubing, and complex slurry pump components across oil, gas, and geothermal sectors.
Ion Vapor Deposition (IVD)
IVD is a hybrid process where the coating material—most commonly aluminum—is evaporated and ionized in an argon plasma, then attracted to the negatively biased substrate. It provides non-line-of-sight coverage, excellent adhesion on high-strength steels and titanium alloys, and was originally developed as a non-toxic alternative to cadmium plating in aerospace applications.
IVD performance profile:
- Coating thickness: 8–50 microns achievable depending on class specification
- No hydrogen embrittlement risk: vacuum process generates no hydrogen
- Continuous service temperature: up to 500°C (925°F)
- Applications: aerospace fasteners, structural components, fatigue-critical parts
MIL-DTL-83488D governs IVD aluminum specifications with three classes based on thickness requirements:
- Class 1: 0.0010 inch (25.4 µm) minimum for general corrosion protection
- Class 2: 0.0005 inch (12.7 µm) for parts with dimensional tolerance limitations
- Class 3: 0.0003 inch (7.6 µm) for close tolerance applications like threaded fasteners
IVD aluminum is typically treated with supplementary chromate conversion coating (Type II) to maximize corrosion resistance and paint adhesion. Documented aerospace applications include fatigue-critical wing skins, bulkheads, landing gear components, and millions of threaded fasteners.

Key Factors That Affect Thin Film Deposition Performance on Metals
Coating performance depends on precise control of input and process variables. The most influential factors include:
Substrate material and condition:
- Alloy composition affects diffusion kinetics—chromium and nickel in stainless steels retard boron diffusion
- Surface roughness determines nucleation uniformity; PVD increases Ra by only ~0.02 µm for sputtering/PACVD
- Oxide layer formation during heating can prevent adhesion if not removed
- Prior heat treatment state must be preserved; coating temperature must stay below tempering temperature
Process conditions inside the chamber are equally critical to the final result.
Process parameters:
- Deposition temperature directly affects coating uniformity, phase composition, and hardness
- Chamber pressure influences mean free path and coating density
- Gas composition determines reaction chemistry and stoichiometry
- Dwell time controls coating thickness and diffusion depth
Part geometry introduces constraints that determine which deposition method is even viable.
Part geometry and size:
- Wall thickness affects thermal mass and heating uniformity
- Bore depth and aspect ratio determine whether line-of-sight or conformal methods are appropriate
- Overall part dimensions must fit within available furnace or chamber volume
- CVD diffusion coating can handle large-diameter components (up to 68+ inches) where PVD cannot
Finally, what happens after deposition is just as important as the process itself.
Post-treatment operations:
- IVD coatings require chromate conversion or sealing to reduce porosity
- Some CVD coatings benefit from post-deposition burnishing
- Skipping required post-treatment steps compromises final coating quality and corrosion resistance
Common Misconceptions About Thin Film Deposition for Metals
Misconception: Thinner Coatings Mean Weaker Protection
In diffusion-based processes, hardness and corrosion resistance derive from the intermetallic compound formed at depth in the substrate, not from coating thickness alone. In ASTM G65 dry sand rubber wheel abrasion tests, thin submerged-arc surfacing Fe-Cr-C layers showed over seven-fold reduction in volume loss compared to thicker arc-sprayed nickel-based coatings.
A 25-micron diffusion coating can outperform a much thicker thermal spray coating in adhesion and wear resistance. The metallurgical bond eliminates the micro-cracking, spallation, and delamination that plague thick mechanical overlays under high point-loading or thermal expansion mismatch.
Misconception: All Vapor Deposition Methods Are Interchangeable
Engineers often specify "PVD" or "CVD" as a generic requirement without understanding that the method must match the performance need:
- PVD for precision hardness on tool surfaces and tight-tolerance components
- CVD diffusion for wear/corrosion resistance on large industrial components with complex geometries
- IVD for corrosion protection on aerospace structural alloys where cadmium is restricted

Using the wrong method by default is a common and costly mistake. A line-of-sight PVD coating on a pump impeller with complex internal passages will leave critical surfaces unprotected. Conversely, specifying CVD diffusion coating for a precision ground die insert may introduce dimensional changes that require post-coating grinding.
Misconception: Vapor-Phase Films Are Only for Small Parts
While thin film deposition has high visibility in semiconductor and optics applications, the same fundamental vapor-phase chemistry has been applied to industrial metal components for decades. Scale is rarely the limiting factor it once was:
- CVD processes coat 24-foot heat exchanger tubes in petrochemical plants
- Thermal diffusion boronizing covers large spools, multi-meter tubing, and complex slurry pump components across oil, gas, and geothermal sectors
- Diffusion coating is now viable for pump housings, valve bodies, and heat exchanger components at industrial scale
VaporKote's large-capacity industrial furnaces handle components up to 68 inches in diameter — a practical demonstration of how far commercial reactor scaling has advanced these technologies for heavy industry.
Conclusion
Thin film deposition for metals encompasses several distinct vapor-phase surface engineering methods—PVD, CVD/diffusion coating, and IVD—each capable of transforming a metal surface's wear and corrosion behavior without altering the component's dimensional integrity.
The difference between a coated component that fails early and one that doubles or triples service life depends on matching the right deposition method to the substrate, operating environment, and geometry. Each method has a clear domain:
- PVD delivers extreme hardness and tight dimensional control, but line-of-sight limitations confine it to simpler external geometries
- CVD diffusion coating provides metallurgical bonding and conformal coverage, making it the preferred choice for components with internal passages, recessed features, or large formats
- IVD aluminum is the aerospace standard for corrosion protection where cadmium is restricted and hydrogen embrittlement must be avoided
The most cost-effective strategy is straightforward: specify the deposition method based on your component's failure mode, geometry constraints, and operating environment—not on familiarity or lowest initial cost.
Frequently Asked Questions
Which is better, CVD or PVD?
The right choice depends on the application. CVD — particularly thermal diffusion processes like boronizing — suits industrial components with complex geometries (pump internals, valve bodies) where conformal coverage and deep adhesion matter. PVD is better for precision tooling such as cutting tools or dies, where ultra-thin, line-of-sight coatings with tight thickness control are the priority.
What is the difference between CVD and ALD?
CVD deposits material through continuous chemical reactions with multiple reactants present simultaneously. ALD (Atomic Layer Deposition) instead builds coatings one atomic layer at a time using self-limiting sequential reactions, offering nanometer-scale precision — but at slower throughput and thinner build-up, making it less practical for industrial metals that need durable coatings of 10+ microns.
How long does PVD coating last?
PVD coating lifespan varies widely depending on coating material, substrate, and operating environment. On cutting tools, PVD coatings like TiN typically extend tool life 2–5x compared to uncoated equivalents. In high-wear or high-temperature industrial environments, coating longevity depends heavily on film thickness, substrate preparation, and whether the base metal received any pre-treatment such as thermal diffusion coating.