
Introduction
Aluminizing is a thermochemical diffusion process that impregnates aluminum into the surface of a base metal, creating a metallurgically bonded intermetallic layer rather than an applied surface coating.
In petrochemical, oil and gas, aerospace, and manufacturing operations, it serves as a frontline defense against component failure in high-temperature, corrosive environments. When furnace tubes, heat exchangers, and downhole equipment face temperatures exceeding 500°C alongside sulfur-bearing gases and carburizing atmospheres, unprotected metals degrade rapidly. The resulting unplanned shutdowns can exceed $1 million per hour in large refineries.
This article covers the precise definition of aluminizing, the step-by-step process mechanics, the industries that depend on it, and the critical variables that determine coating performance and service life.
TL;DR
- Aluminizing drives aluminum into the base metal surface through heat, forming a bonded intermetallic layer—not a removable film
- Coated surfaces resist oxidation, sulfidation, carburization, and hydrogen permeation in harsh operating environments
- Common methods include pack cementation, vapor phase aluminizing (VPA), and chemical vapor deposition (CVD)
- Coating depth and uniformity depend on processing temperature, dwell time, base metal composition, and powder formulation
- Operationally preferred in oil refining, petrochemicals, and aerospace where surface failure triggers expensive downtime
What Is Aluminizing?
Aluminizing is a thermochemical diffusion treatment in which aluminum is introduced into the surface layer of a metal substrate—typically steel, nickel, or cobalt-based alloys—through elevated-temperature processing. The process forms an aluminum-rich intermetallic compound that bonds directly to the base metal at the atomic level. According to ASTM B875, this creates a metallurgically bonded interdiffusion zone that chemically alters the substrate surface.
The result: the surface acquires aluminum's superior corrosion and oxidation resistance while the base metal retains its original structural strength, rigidity, and high-temperature mechanical properties. Unlike aluminum alloys where the entire cross-section is changed, aluminizing modifies only the surface zone—typically 25 to 127 micrometers deep depending on substrate class.
During processing, aluminum atoms diffuse into the base metal lattice rather than simply adhering to the surface. This diffusion mechanism produces an intermetallic layer with key advantages over conventional surface coatings:
- Cannot be removed without machining — bonded at the atomic level, not deposited on top
- Outperforms electroplating and hot-dip galvanizing under thermal cycling
- Resists delamination and spalling that affect separately deposited metallic layers
How the Aluminizing Process Works
Aluminizing converts the surface chemistry of a metal component by exposing it to aluminum-bearing vapors or powders at high temperatures (typically 700–1100°C), triggering inward diffusion of aluminum atoms to create a graded intermetallic zone. The process requires five core inputs:
- Base metal component to be treated
- Aluminum-containing powder or vapor as the diffusion source
- Halide salt activator (such as NH₄Cl or AlF₃) that generates metal halide vapor
- Inert filler (typically aluminum oxide) to prevent sintering
- Controlled heat environment to drive and sustain diffusion
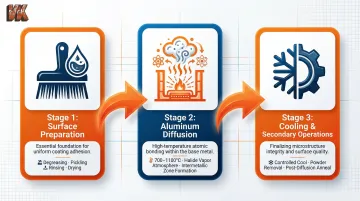
Surface Preparation
Surface cleanliness is critical. Components undergo thorough degreasing, pickling (acid cleaning), rinsing, and drying to remove oils, oxides, scale, and contaminants. Any contamination disrupts the diffusion reaction and results in uneven or defective coatings. This preparation step determines whether the subsequent diffusion will be uniform across complex geometries.
Aluminum Diffusion
The component is packed in an airtight retort surrounded by the aluminum-bearing powder mixture, then heated in a furnace to process temperature. The activator vaporizes and creates aluminum halide gas (AlCl₃, AlF₃, or similar compounds) that migrates to the component surface, where it decomposes and deposits aluminum. The aluminum then diffuses into the substrate, forming intermetallic phases such as Fe₂Al₅, FeAl, or β-NiAl depending on the base metal composition.
Because the diffusion profile varies by substrate and end-use requirement, VaporKote formulates on-site powder mixes for each application—adjusting the ratio of aluminum powder, activator, and inert filler to control coating uniformity, thickness, and composition.
Cooling and Secondary Operations
After the soak period (typically 2 to 24 hours depending on desired case depth), the retort is cooled in a controlled manner and the component is removed and cleaned of excess powder. A post-diffusion anneal or heat treatment may follow to recover the substrate's mechanical properties and stabilize the diffusion zone. The resulting aluminized layer contains a minimum of approximately 20% aluminum by weight, with ASTM B875 mandating that the outer 15% of the coating must contain at least 28% aluminum.
Why Industries Rely on Aluminizing
Aluminizing addresses four specific corrosion mechanisms simultaneously, making it indispensable in severe service environments:
Oxidation resistance: Aluminum forms a continuous α-Al₂O₃ (alumina) scale—a self-sealing, chemically stable barrier that grows slowly and remains adherent even under thermal cycling. This alumina scale is significantly more stable than chromia scales, particularly in combustion environments containing water vapor.
Sulfidation resistance: In sulfur recovery units and refining environments above 590°C, aluminized stainless steel exhibits considerably lower sulfidation rates than bare stainless steel. The aluminum oxide barrier prevents hydrogen sulfide from dissociating at the metal surface.
Carburization resistance: The alumina layer is unreactive to carbon, preventing the formation of metal carbides that cause catastrophic metal dusting in high-hydrocarbon environments. Case studies document aluminized burner plates in steam reformers more than doubling service life under severe metal dusting conditions.
Hydrogen permeation resistance: Recent studies demonstrate that iron aluminide coatings form a Fe₂Al₅/Fe₃Al intermetallic structure that acts as a near-perfect hydrogen permeation barrier, preventing hydrogen embrittlement in clean energy infrastructure.

Without aluminizing, unprotected components are exposed to a cascade of failure modes:
- Metal dusting in carburizing atmospheres
- Scaling and surface degradation from oxidation
- Sulfidation attack on furnace tubes and heat exchangers
- Premature failure leading to unplanned shutdowns
In large refineries, unplanned outages cost up to $1 million per hour. That cost exposure alone justifies aluminizing as a standard protective measure, not an afterthought.
Aluminizing is operationally preferred in industries that adhere to engineering standards such as ASTM, ASME, SAE, and API. API RP 571 explicitly recognizes aluminum diffusion treatments as beneficial in preventing metal dusting and reducing sulfidation rates. For certain high-temperature service components—furnace tubes, reformer tubes, heat exchanger internals—it functions as a best-practice specification requirement that engineers build into design from the start.
Aluminizing a component costs a fraction of replacing it or sourcing premium exotic alloys like Inconel throughout. Aluminized carbon and low-alloy steels outperform bare 300-series stainless steels in environments above 500°C, allowing engineers to apply aluminum's protective properties at the surface without sacrificing the strength and machinability of the base metal.
Where Aluminizing Is Applied
Aluminizing is most commonly applied to components and systems facing extreme thermochemical stress:
- Furnace tubes and reformer tubes in petrochemical and refining operations
- Heat exchanger tubes and internals exposed to high-temperature corrosive fluids
- Downhole tubing and production vessels in oil and gas extraction
- Catalyst trays in chemical processing plants
- Exhaust manifolds and turbine components in aerospace and power generation
- Wire screens and piping in sulfur recovery and chemical processing plants
- Agricultural or industrial parts exposed to abrasive or corrosive media
Component size is rarely a limiting factor. VaporKote's furnaces handle parts up to 68 inches in diameter, covering standard and oversized industrial components — including large-diameter heat exchanger shells and pressure vessel components.
Aluminizing is typically specified at the design/procurement stage for components with known exposure to temperatures above 500°C, sulfur-containing gases, carburizing atmospheres, or high-humidity corrosive environments. It may also be specified as a refurbishment or life-extension measure when uncoated components show early wear patterns.
Aluminizing is primarily a one-time treatment performed before a component enters service, since the diffusion layer becomes part of the metal itself. However, if a component is refurbished or machined down, the process may be reapplied to restore surface protection.
Key Factors That Affect the Aluminizing Process
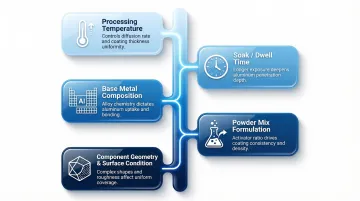
Five process variables have the most direct impact on coating quality, depth, and performance:
Processing temperature — Higher temperatures accelerate diffusion and increase coating depth, but can cause grain growth, carbide precipitation, or substrate distortion. The optimal range is typically 700–1100°C depending on the base metal and target case depth. Because the diffusion coefficient follows an Arrhenius relationship, temperature has an exponential effect on diffusion rate — small shifts matter.
Soak/dwell time — Longer exposure produces deeper aluminum diffusion and higher surface aluminum content. Coating thickness increases parabolically with time, following x ∝ √Dt. Over-aluminizing is a real risk; excessive dwell time can introduce brittleness that undermines coating performance.
Base metal composition — Substrate alloying elements — chromium, nickel, iron — govern how aluminum diffuses and which intermetallic phases form. In nickel-based superalloys, outward nickel diffusion forces less mobile elements like W, Mo, Cr, and Ti to concentrate in the interdiffusion zone, creating precipitate phases with distinct mechanical properties. Each alloy system requires its own process parameters.
Powder mix formulation — The ratio of aluminum powder, activator, and inert filler controls aluminum vapor activity, directly shaping coating uniformity, thickness, and composition. On-site formulation control — rather than off-the-shelf mixes — is what enables consistent results across varied geometries and part sizes.
Component geometry and surface condition — Blind holes, thick cross-sections, and complex geometries create uneven diffusion paths if packing is inadequate. Surface cleanliness matters equally: residual contamination produces localized defects that compromise coating integrity from the start.
Common Misconceptions About Aluminizing
A few persistent misunderstandings lead engineers to misapply aluminizing or overlook it entirely. Here's what the process actually does — and doesn't — do.
Aluminizing Creates a Diffusion Zone, Not a Surface Layer
The most frequent misunderstanding is equating aluminizing with applying an aluminum coating on top of the metal, like paint, plating, or thermal spray. The aluminum actually diffuses into and bonds with the base metal at the atomic level, creating an intermetallic compound. Per ASTM B875, this metallurgically bonded interdiffusion zone cannot peel, flake, or delaminate under thermal cycling the way surface coatings can.
Hot-Dip vs. Diffusion Aluminizing
These two methods share a name but produce very different results:
| Hot-Dip Aluminizing | Diffusion Aluminizing (Pack/CVD) | |
|---|---|---|
| Method | Steel immersed in molten aluminum | Aluminum vapor diffused at elevated temperature |
| Resulting Layer | Brittle Fe₂Al₅ layer + unalloyed aluminum overlay | Deeply integrated intermetallic zone |
| Best For | Sheet metal, structural applications | High-temperature, high-corrosion industrial components |
| Integration Depth | Shallow | Deep atomic bonding with base metal |
Choosing the wrong method for a given application — say, using hot-dip aluminized sheet in a high-temperature gas turbine environment — leads to premature coating breakdown.
Aluminizing Has Specific Corrosion Limits
While aluminizing provides excellent resistance to oxidation, sulfidation, carburization, and hydrogen permeation, it does not protect against all forms of corrosion equally. Aluminide coatings fail rapidly in specific molten alkali salts (such as Na₂SO₄-K₂SO₄ eutectic melts) (low-melting salt mixtures) and aggressive chlorine-bearing atmospheres where aluminum itself is attacked. For waste incineration, molten salt reactors, or chlorine-rich chemical processing environments, an alternative coating system — or a hybrid approach — may be the more reliable choice.
Frequently Asked Questions
What does aluminized mean?
"Aluminized" refers to a metal that has undergone the aluminizing process, meaning aluminum has been diffused into its surface to form a protective intermetallic layer. This treatment improves corrosion, oxidation, and heat resistance for high-temperature industrial applications.
What is the difference between aluminizing and galvanizing?
Galvanizing applies a zinc coating (typically by hot-dipping) to protect steel from corrosion at ambient temperatures. Aluminizing uses aluminum to create a diffusion layer suited for high-temperature and chemically aggressive environments. The two processes address different service conditions and temperature ranges.
What metals can be aluminized?
Aluminizing can be applied to all wrought and cast steels—plain carbon, low-alloy, ferritic, austenitic, and highly alloyed grades—as well as nickel-based and cobalt-based alloys. The process parameters are adjusted based on substrate composition.
What is better, aluminized steel or stainless steel?
The choice depends on the application. Aluminized steel outperforms stainless in high-temperature oxidation and sulfidation resistance at a lower cost, while stainless steel offers broader corrosion resistance in aqueous or chloride environments where aluminum would be attacked.
How long does an aluminized coating last?
Because the aluminized layer is metallurgically bonded to the base metal, it does not delaminate over time. Service life depends on operating severity, but documented case studies show aluminized components exceeding 10 years of continuous operation in severe metal dusting conditions.
Is aluminized steel healthy to cook with?
Aluminized steel used in consumer cookware is considered safe under normal cooking temperatures; the aluminum oxide layer is chemically stable and does not leach at normal cooking temperatures. Industrial aluminizing operates at far higher temperatures and serves entirely different applications than consumer cookware.


