
Introduction
Aluminum and stainless steel are workhorses of industrial design—but pairing them without proper precautions triggers an electrochemical reaction that steadily degrades the aluminum. Heat exchangers, oil rigs, aerospace assemblies, and processing equipment all face this electrochemical threat. While both metals resist corrosion individually, combining them without proper precautions accelerates aluminum breakdown, leading to premature component failure, costly downtime, and safety risks.
The global cost of corrosion reaches $2.5 trillion annually, equivalent to 3.4% of global GDP. The U.S. Department of Defense alone spends $20.8 billion each year preventing and mitigating corrosion on military equipment.
In offshore operations, where aluminum-stainless steel assemblies are common, corrosion accounts for up to 60% of maintenance costs—and offshore maintenance runs 15 to 20 times more than equivalent onshore work.
This guide covers the root causes, consequences, and proven prevention strategies for galvanic corrosion between aluminum and stainless steel.
TLDR
- Galvanic corrosion occurs when aluminum and stainless steel make electrical contact in the presence of a conductive electrolyte—aluminum corrodes first
- Severity depends on potential difference, electrolyte presence, and surface area ratio between metals
- Aluminum-stainless steel is a high-risk pairing—the two metals sit far apart on the galvanic series
- Prevention includes electrical isolation, protective coatings, favorable area ratios, and careful material selection
- Harsh environments demand routine inspection, drainage design, and diffusion-bonded coatings for lasting protection
What Causes Galvanic Corrosion Between Aluminum and Stainless Steel?
Galvanic corrosion is an electrochemical process that occurs when two dissimilar metals with different electrode potentials are electrically connected in the presence of a conductive electrolyte. The less noble metal (aluminum) oxidizes and deteriorates while the more noble metal (stainless steel) remains protected.
The reaction requires three simultaneous conditions. Eliminate any one of them and corrosion stops — which gives engineers a clear target for intervention.
Electrochemical Potential Difference
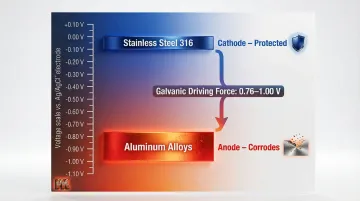
Aluminum sits significantly lower on the galvanic series than stainless steel, creating a large voltage difference between the two metals. In seawater, aluminum alloys range from -0.76 to -1.00 V (vs Ag/AgCl), while passive stainless steel 316 ranges from -0.00 to -0.10 V. This 0.76 to 1.00 V potential difference creates a strong driving force for corrosion.
This potential difference drives electrons from aluminum (anode) to stainless steel (cathode), dissolving aluminum ions into the surrounding electrolyte. The result is the familiar white powdery deposit — aluminum oxide — at and around the contact zone.
Presence of an Electrolyte
The electrolyte — any conductive fluid including water, saltwater, process fluids, condensation, or industrial cleaning solutions — completes the circuit and allows ion transfer between the two metals. Without this conductive bridge, the galvanic cell cannot operate.
Industrial environments are particularly electrolyte-rich:
- Offshore platforms face constant saltwater exposure
- Chemical processing plants handle corrosive process fluids
- Heat exchangers operate with water-based coolants
- Condensation forms in temperature-cycling equipment
Electrical Contact and Unfavorable Area Ratios
Any electrical connection — direct metal-to-metal contact or an indirect path through conductive fasteners — is sufficient to sustain the galvanic cell.
The area ratio problem is critical: When a small aluminum component (a fitting or fastener) couples to a large stainless steel surface, the current density concentrated on the small anode is extremely high, causing accelerated and localized destruction. Research confirms that galvanic current density at the anode scales directly with the cathode-to-anode area ratio.
NAVAIR testing demonstrated that bare SS316 fasteners installed in AA7075-T6 panels caused localized pitting 280–370 µm deep after 504 hours of salt spray exposure. Reversing the ratio — large aluminum surface, small stainless fasteners — reduces current density at the anode and slows the attack, though it does not eliminate the risk entirely.
What Happens When Galvanic Corrosion Goes Unaddressed?
Aluminum components lose cross-section through pitting and surface dissolution, weakening joints, threads, and load-bearing elements. In industrial settings this means:
- Heat exchanger tube sheet failures
- Loosened pipe fittings
- Compromised aerospace structural brackets
- Thread stripping in fasteners
- Joint movement and structural instability
Corrosion-driven failures lead to unplanned downtime, emergency replacement costs, and in safety-critical industries like oil & gas and aerospace, potential regulatory violations and liability exposure. With offshore maintenance costing 15 to 20 times more than onshore work, an unchecked galvanic pair can turn a $50 fastener problem into a six-figure shutdown.
Warning Signs You Are Experiencing Galvanic Corrosion
These signs point to active galvanic corrosion:
- White or gray powdery deposits (aluminum oxide) forming around contact points, especially near fasteners or brackets
- Pitting, cratering, or surface thinning on aluminum at the junction with stainless steel — while the stainless remains unaffected
- Loosening fasteners, thread stripping, or joint movement in aluminum parts fitted with stainless steel hardware, indicating material loss is already underway
How to Prevent Galvanic Corrosion Between Aluminum and Stainless Steel
Prevention works best as a layered approach: design decisions made before assembly, physical barriers and surface treatments applied during installation, and ongoing inspection throughout service life.
Use Electrical Isolation at Contact Points
Install non-conductive insulating materials (neoprene, PTFE, or nylon washers, gaskets, and sleeves) between aluminum and stainless steel surfaces to physically break the electrical circuit required for galvanic corrosion.
Critical requirements:
- Specify non-absorbent materials only—porous or water-retaining insulators trap electrolyte at the joint, potentially accelerating corrosion
- Apply this measure at every contact point: under bolt and screw heads, on shanks passing through aluminum, and at flange faces
- Any uninsulated point of contact can sustain the galvanic cell
MIL-STD-889D mandates complete isolation using dielectric sleeves (Mylar/G-10) and washers (PTFE/Neoprene) at all contact points. Partial isolation fails because galvanic cells remain active if bolt shanks touch the bore.
Recommended isolation materials:
| Material | Water Absorption | Strength | Best Application |
|---|---|---|---|
| G-10 Epoxy Glass | 0.10% | 50,000 psi compressive | High-pressure flanges, washers |
| Mylar | 0.80% | High dielectric | Bolt sleeves |
| PTFE (Teflon) | Minimal | Chemically stable to 600°F | Aggressive chemical environments |

Apply Protective Coatings to Create a Barrier
Apply corrosion-resistant coatings to one or both metal surfaces to interrupt the electrical path and shield the base metal from the electrolyte. Options range from epoxy primers and anodizing on aluminum to industrial coatings on stainless steel components.
Critical principle: Always coat the stainless steel (cathode) first, or both metals. Coating only the aluminum (anode) creates severe risk of rapid localized pitting at coating defects (holidays) due to concentrated galvanic currents. In testing, applying coating to SS316 fasteners reduced corrosion damage in aluminum by 84%.
For industrial-grade protection in demanding environments (high temperatures, corrosive process fluids, mechanical wear), metallurgically bonded diffusion coatings offer a step up from paint or plating. Chemical vapor deposition (CVD) processes form an intermetallic compound layer at the metal surface itself, not just on top of it.
This bond-level adhesion is what sets diffusion coatings apart in abrasive or thermally cycled service conditions where surface-only coatings delaminate or undercut.
In high-temperature environments like 600°C recovery boilers, organic paints and electroplating fail, whereas CVD thermal diffusion coatings (aluminizing/boronizing) maintain integrity and prevent galvanic coupling. VaporKote's aluminizing process, for instance, creates a protective aluminum layer on stainless steel components that has demonstrated corrosion resistance at service temperatures where conventional coatings break down.
Surface-only coatings (paint, powder coat) serve as a first line of defense but are susceptible to holiday defects, abrasion, and undercutting. Industrial applications should pair surface coatings with mechanical isolation for redundant protection.
Design for Favorable Surface Area Ratios
Where mixed-metal assemblies cannot be avoided, design so that the stainless steel component is smaller in surface area relative to the aluminum component. This distributes galvanic current over a larger anode, reducing the corrosion rate significantly.
Avoid the inverse configuration: Do not use aluminum fasteners, rivets, or small fittings on large stainless steel structures. The concentrated current on the small aluminum anode will cause rapid destruction of the fastener.
Select Compatible Metals or Use Intermediate Materials
Where design flexibility exists, replace one of the metals with a material closer to the other on the galvanic series to reduce the electrochemical potential difference and slow the galvanic driving force.
Use zinc-based sacrificial anodes in environments where the aluminum component must be protected and electrical isolation is impractical. Zinc (-0.98 to -1.03 V vs Ag/AgCl) is more active than both aluminum (-0.76 to -1.00 V) and stainless steel in seawater, making it an effective sacrificial anode.
DNV-RP-B401 specifically recommends zinc anodes for closed compartments, as aluminum anodes risk acidification and hydrogen gas production in water-flooded spaces without ventilation.
Best Practices for Long-Term Prevention and Control
Implement scheduled inspection intervals for all mixed-metal assemblies, focusing on known contact points. Document baseline condition at installation so that progressive material loss can be identified early before structural integrity is compromised.
Inspection protocols:
- API 570 (Piping Inspection Code) requires external visual inspections at maximum 5-year intervals or half the remaining pipe life, whichever is shorter
- API 510 (Pressure Vessel Inspection Code) requires internal inspections at maximum 10-year intervals or half the remaining life
- Look for early indicators (white deposits, surface pitting) at each inspection
- Replace insulating components (washers, gaskets) that show signs of degradation, compression set, or moisture intrusion

Design and maintain proper drainage in assemblies to minimize electrolyte pooling at aluminum-stainless interfaces. Slope surfaces away from joints, avoid crevices that trap water, and select sealants compatible with both metals to prevent moisture ingress. Physical design choices here directly reduce how often aggressive-environment protocols become necessary.
For aggressive environments:
- In offshore, marine, or high-humidity industrial environments, increase inspection frequency
- Apply corrosion-inhibiting compounds or sealants at joints during routine maintenance shutdowns
- Seal all faying surfaces and edges to preclude liquid entrance, eliminating the electrolyte entirely
These environmental controls only work if the people installing and maintaining assemblies understand the risks. Train personnel to recognize galvanic risk configurations — particularly the hazard of substituting aluminum fasteners for stainless in field repairs, or introducing a new electrolyte source (such as a process fluid leak) at an existing mixed-metal joint.
For critical components in petrochemical, aerospace, or oil and gas applications, work with a coating and materials specialist who adheres to ASTM, ASME, SAE, and API standards. The right combination of material selection, coating system, and inspection protocol depends heavily on the specific service environment — there's no one-size-fits-all answer.
VaporKote's engineering team works directly with industrial clients to assess mixed-metal assembly risk and recommend diffusion coating strategies suited to the actual operating conditions.
Conclusion
Galvanic corrosion between aluminum and stainless steel is predictable and preventable. It requires three specific conditions—electrochemical potential difference, electrical contact, and an electrolyte—and interrupting any one of them through isolation, protective coatings, smart design, or material selection is enough to stop or significantly slow the reaction.
Proactive prevention at the design and installation stage costs far less than reactive repairs, emergency downtime, or component replacement. In offshore environments, where maintenance runs 15–20 times the cost of onshore work and corrosion accounts for roughly 60% of total maintenance spend, getting this right upfront matters.
Protective coatings—including thermal diffusion processes like aluminizing—add another layer of defense by modifying the surface chemistry of base metals, reducing their electrochemical reactivity in mixed-metal assemblies. For operations where long service life and minimal downtime aren't optional, that kind of protection pays for itself. VaporKote's aluminizing services are engineered specifically for these demanding environments.
Frequently Asked Questions
Will aluminum and steel corrode together?
Yes. When electrical contact, an electrolyte, and a sufficient electrochemical potential difference are all present simultaneously, aluminum acts as the anode and corrodes preferentially. With proper isolation and protective measures, the two metals can be used together safely.
Can you put steel and aluminum together?
Yes, but precautions are required: isolate contact points with non-conductive washers or gaskets, apply protective coatings, and maintain favorable surface area ratios. Skip these steps and galvanic corrosion of the aluminum is likely, particularly in wet or saline environments.
What are the three conditions required for galvanic corrosion to occur?
Three conditions must exist at the same time: dissimilar metals with different electrode potentials, electrical contact between them, and a conductive electrolyte — water, saltwater, or a process fluid — bridging both surfaces. Remove any one condition and the galvanic cell cannot operate.
Which metal corrodes when aluminum and stainless steel are in contact?
Aluminum is always the metal that corrodes in this pairing because it is the more anodic (less noble) metal. It acts as the anode, giving up metal ions into the electrolyte, while the stainless steel acts as the cathode and remains largely unaffected.
How does surface area ratio affect galvanic corrosion between these metals?
A small aluminum component coupled to a large stainless steel surface is the worst-case scenario: galvanic current concentrates on a small anode, sharply accelerating aluminum loss. A large aluminum surface with small stainless fasteners carries comparatively lower risk, though not zero risk.
Are protective coatings effective at preventing galvanic corrosion on industrial components?
Coatings are a proven prevention method, but effectiveness depends on coating type and application quality. For industrial environments with high temperatures, mechanical wear, or aggressive process fluids, metallurgically bonded diffusion coatings (applied via CVD processes) offer superior long-term protection compared to paint or plating, which can chip or undercut over time.


