
Introduction
Engineers and procurement professionals selecting materials for aggressive industrial environments face a critical challenge: aluminum's corrosion resistance is conditional, not absolute. While many specify aluminum assuming universal protection, the reality is more nuanced. Approximately 40% of premature aluminum component failures in industrial settings stem from mismatched alloy selection or inadequate surface treatment for the operating environment.
The core tension lies in aluminum's self-protecting oxide layer. This barrier offers genuine resistance in neutral pH environments but breaks down rapidly in chloride-rich seawater, acidic process fluids, or when coupled with dissimilar metals. A 5083 marine alloy can last decades in saltwater while a 7075 aerospace alloy may fail within months under identical exposure — the alloy choice is that consequential.
This article explains how aluminum's passive protection works, what environmental factors undermine it, and what concrete steps — from alloy selection to advanced diffusion coatings — can extend corrosion performance in demanding industrial conditions where unplanned replacements and downtime directly impact production schedules and budgets.
TL;DR
- Aluminum forms a 4-6 nanometer oxide layer that self-heals when damaged, offering natural corrosion resistance in pH 4–8.5 environments
- Chloride exposure, galvanic coupling, temperatures above 65°C, and strong acids or alkalis all break down this natural protection
- 5xxx series alloys (5052, 5083) lead in marine environments; 6061 balances strength with protection
- 2xxx and 7xxx series alloys sacrifice corrosion resistance for strength and need surface treatment in aggressive conditions
- Anodizing, conversion coatings, and thermal diffusion processes significantly extend protection in industrial-grade applications
- Effective corrosion management combines alloy selection, environmental analysis, and surface treatment — no single factor works alone
How Aluminum's Natural Corrosion Resistance Works
When aluminum contacts oxygen or water, it instantly forms a thin aluminum oxide (Al₂O₃) layer measuring 4-6 nanometers thick. Unlike iron rust, which flakes off and exposes fresh metal to continued attack, this oxide film is dense, adherent, and non-porous—effectively sealing the substrate from further oxidation. If mechanically scratched or damaged, the layer reforms within 20 minutes in ambient conditions, giving aluminum a self-healing capability that most structural metals lack.
This passive barrier is stable only within a specific pH range. According to the Pourbaix diagram for aluminum-water systems, the oxide layer maintains integrity between approximately pH 4 and 8.5. Within this window, aluminum resists general corrosion effectively.
Outside that range, the oxide dissolves into water-soluble species—aluminum ions (Al³⁺) in acidic conditions below pH 4, and aluminate ions (AlO₂⁻) in alkaline environments above pH 9—leaving bare metal vulnerable to rapid uniform attack.
How alloying affects the oxide layer:
- 1xxx series (pure aluminum): forms the most stable, uniform oxide with excellent chemical resistance
- 5xxx series (magnesium additions): maintains oxide stability while improving strength, particularly in marine environments
- 2xxx and 7xxx series (copper and zinc additions): alters surface electrochemistry, creating micro-galvanic cells that reduce oxide effectiveness and increase susceptibility to localized corrosion

Corrosion resistance vs. immunity:
Aluminum is corrosion resistant, not corrosion immune. The passive oxide provides a solid baseline defense, but aggressive environments—concentrated acids, caustic process fluids, chloride-saturated marine atmospheres—can overwhelm it. In oil & gas, petrochemical, and offshore applications, that difference determines whether bare aluminum holds up or requires supplemental coating protection.
What Affects Aluminum's Corrosion Resistance in Real-World Environments
Several factors determine whether aluminum's natural oxide protection holds in service. These variables can act independently or combine to accelerate degradation:
Chloride ion exposure:
Chlorides found in seawater, industrial brines, and coastal atmospheres are particularly aggressive. These negatively charged ions penetrate the oxide layer at localized weak points, initiating pitting corrosion. Once a pit forms, hydrolysis of dissolved aluminum ions drives the local pH down to highly acidic levels (0.85–2.3), preventing oxide reformation and allowing the pit to propagate deep into the metal.
In seawater immersion, corrosion rates range from 0.016 mm/year for resistant alloys like 5083 to 0.10 mm/year for more vulnerable grades.
Galvanic corrosion risk:
When aluminum contacts a more noble metal—such as stainless steel, copper, or brass—in the presence of an electrolyte, aluminum acts as the sacrificial anode and corrodes preferentially.
The severity scales with the cathode-to-anode area ratio. A large stainless steel plate with small aluminum fasteners concentrates galvanic current onto the fasteners, causing rapid perforation. Aluminum plates with stainless fasteners distribute the effect over a wider area, resulting in milder surface degradation.
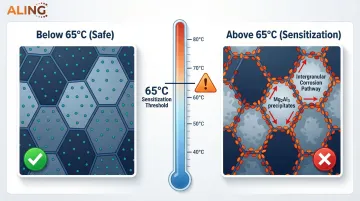
Temperature effects:
Of greater concern, 5xxx series alloys containing more than 3.5% magnesium (such as 5083 and 5456) undergo sensitization when exposed to temperatures above 65°C (150°F) for prolonged periods. At these temperatures, magnesium precipitates as anodic Mg₂Al₃ particles along grain boundaries, creating pathways for catastrophic intergranular corrosion.
Common Types of Corrosion That Affect Aluminum
Pitting corrosion:
Localized attack where the oxide layer breaks down in small spots, creating deep cavities that compromise structural integrity without visible surface degradation. Pitting is most common in chloride-rich environments and is particularly dangerous because extensive subsurface damage can occur while the surface appears largely intact.
Crevice corrosion:
Occurs in confined spaces—joints, fastener holes, lap joints, unsealed gasket areas—where stagnant electrolyte accumulates. Oxygen depletion in these restricted geometries destabilizes the oxide layer locally, while chloride concentration and acidification create a highly corrosive micro-environment. This is a critical consideration in fabricated assemblies and multi-component systems.
Intergranular corrosion:
Affects certain alloys (notably 2xxx and some 7xxx series) where grain boundary regions become anodic relative to the surrounding aluminum matrix. This leads to preferential attack along grain boundaries, causing crack propagation with serious structural implications.
In aerospace and load-bearing applications, this mechanism can progress to stress corrosion cracking (SCC)—a failure mode where corrosion, tensile stress, and susceptible microstructure act together to cause sudden fracture.
How to Enhance Aluminum's Corrosion Protection
Aluminum's natural resistance is a starting point, not an endpoint. In aggressive industrial environments—oil & gas facilities, petrochemical plants, marine installations, mining operations—additional surface protection prevents costly component failures and extends service life.
Anodizing
Anodizing artificially thickens the natural oxide layer through an electrochemical process—from a few nanometers to 5–25 microns (Type II sulfuric anodizing) or 12–114 microns (Type III hardcoat anodizing). This improves resistance to pitting and general corrosion while adding significant wear resistance in hardcoat applications.
Alloy compatibility matters here. Anodizing performs best on 1xxx, 5xxx, and 6xxx series alloys, which produce uniform, dense coatings. High-copper 2xxx and high-zinc 7xxx alloys respond poorly, often producing porous, mottled coatings that provide inadequate protection.
Conversion Coatings
Conversion coatings—commonly called chem film or Alodine—are thin chemical films that provide moderate corrosion protection and excellent primer adhesion for subsequent paint systems. Traditional hexavalent chromium (Cr(VI)) coatings offered superior performance, but regulators heavily restrict them due to carcinogenicity. The EU REACH regulation mandates phase-out by 2024. Trivalent chromium process (TCP) coatings and zirconium-based alternatives now provide comparable protection while meeting RoHS and environmental compliance requirements.
Painting and Powder Coating
Paint and powder coating systems physically isolate aluminum from corrosive media. They're particularly effective against galvanic corrosion in multi-metal assemblies and offer broad aesthetic options. That said, coating integrity depends heavily on surface preparation quality—mechanical damage over time remains a real limitation in high-wear environments.
Advanced Diffusion Coatings for Industrial Components
For aluminum parts operating in the most demanding conditions—petrochemical processing, oil drilling, heat exchangers, aerospace—conventional surface treatments may not provide adequate long-term protection. Diffusion coating processes use chemical vapor deposition to form an intermetallic compound at the metal surface, creating a metallurgically bonded protective layer that outperforms conventional treatments in both wear and corrosion resistance.
VaporKote's aluminizing process delivers high-temperature corrosion protection for critical components in oil production, petrochemical refining, and heat exchanger applications. The process adheres to ASTM, ASME, and API standards—providing engineering-grade protection for components where failure carries serious operational and safety consequences.
Because diffusion forms an intermetallic compound bonded directly to the base metal, the resulting layer resists both wear and corrosion at a level conventional coatings can't match. Customers consistently report reduced maintenance costs and extended component life, with documented annual savings reaching into the hundreds of thousands of dollars.

Selecting the Right Aluminum Alloy for Your Application
Corrosion resistance varies dramatically across alloy series. Selecting the right grade requires balancing environmental exposure, mechanical requirements, and fabricability:
| Alloy Series | Primary Element | Corrosion Resistance | Typical Applications |
|---|---|---|---|
| 1xxx | Pure Al (>99%) | Excellent—highest chemical resistance | Chemical processing equipment, piping |
| 3xxx | Manganese | Very good—immune to IGC/SCC | Heat exchangers, storage tanks |
| 5xxx | Magnesium | Excellent in marine/chloride environments | Boat hulls, offshore structures, pressure vessels |
| 6xxx | Mg + Silicon | Good—balanced strength and resistance | Structural extrusions, marine hardware |
| 2xxx | Copper | Poor—susceptible to IGC and pitting | Aerospace (requires cladding/coating) |
| 7xxx | Zinc | Poor—high SCC and exfoliation risk | High-stress aerospace/military components |

5xxx Series for Marine Environments
Alloys 5052 and 5083 are top performers for chloride service. In long-term seawater immersion tests, 5083 exhibits corrosion rates of just 0.016 mm/year, while 5052 shows 0.024 mm/year—both providing decades of reliable service. The key difference: 5052 (2.5% Mg) is immune to thermal sensitization and suitable for temperatures above 65°C, while 5083 (4.0-4.9% Mg) offers higher strength but must be limited to temperatures below 65°C to prevent intergranular corrosion.
6061-T6 vs. 7075: A Common Selection Decision
6061-T6 offers good corrosion resistance with yield strength of 276 MPa (40,000 psi), making it suitable for general structural use and moderate marine exposure. 7075 is significantly stronger at 503 MPa (73,000 psi), but its high zinc content (5.1-6.1%) makes it highly susceptible to stress corrosion cracking in chloride environments.
SCC thresholds for 7075-T6 can drop as low as 220-340 MPa in just 0.5% NaCl solutions. If 7075 must be used in corrosive environments, specify overaged T73 or T7351 tempers—these sacrifice 10-15% strength but restore excellent SCC resistance.
Matching Alloy to Environment
The right alloy balances corrosion resistance, mechanical requirements, fabricability, and operating environment. A 5083 marine alloy and a 7075 aerospace alloy are both aluminum, but their behavior in seawater is dramatically different. Getting alloy selection wrong means relying on coatings and treatments to compensate—adding cost and complexity that a better material choice would have avoided.
Common Misconceptions About Aluminum Corrosion Resistance
Several widely held assumptions about aluminum lead to underprotected components and premature failures in the field. Here are three misconceptions worth addressing directly.
"All aluminum alloys are equally corrosion resistant"
Corrosion resistance varies dramatically across alloy series. A 5083 marine alloy will thrive in seawater where a 7075 aerospace alloy would fail rapidly. Treating alloys interchangeably in material selection—without accounting for alloy-specific corrosion behavior—leads to premature component failures.
"Aluminum doesn't corrode, so it needs no protection"
Aluminum does corrode, particularly through pitting and galvanic mechanisms in chloride-rich or multi-metal environments. The passive oxide layer provides a baseline, not a guarantee. For industrial components with long service life requirements in aggressive environments, additional protection through surface treatment or protective coatings is essential.
"Anodizing is sufficient for all demanding applications"
Anodizing is highly effective for general use, but it has real limitations in extreme temperatures, aggressive chemical environments, and high-wear situations. Engineers specifying aluminum for oil & gas, petrochemical, or heavy industrial applications should evaluate whether more robust protective coating solutions (such as diffusion coatings) are warranted for the specific operational demands of the application.
Frequently Asked Questions
Is aluminum corrosion resistant?
Yes, aluminum is inherently corrosion resistant due to the passive aluminum oxide layer it forms instantly on contact with oxygen. However, the degree of resistance depends heavily on alloy composition, environmental conditions (particularly pH and chloride exposure), and whether supplemental protective treatments have been applied.
How long does it take for aluminum to corrode?
It depends on environment. In mild atmospheric conditions, corrosion rates stay between 0.001–0.01 mm/year, with minimal degradation over decades. In saltwater or industrial settings, localized pitting can begin within months without protective treatment, reaching 0.10 mm/year or higher.
Which aluminum alloys have the best corrosion resistance?
The 5xxx series—particularly 5052 and 5083—offer the best corrosion resistance for marine and harsh environments, thanks to high magnesium content and stable oxide formation. They strike the right balance of strength and corrosion protection that pure 1xxx aluminum can't match.
Is 6061 T6 aluminum corrosion resistant?
Yes, 6061 T6 performs well in atmospheric and freshwater conditions. It's more vulnerable to pitting in saltwater than 5xxx alloys, but with anodizing or conversion coating applied, it's a reliable choice for moderate marine and industrial applications.
Which is more corrosion resistant, 6061 or 7075?
6061 is significantly more corrosion resistant than 7075. The high zinc content (5.1-6.1%) in 7075 reduces its resistance and increases susceptibility to stress corrosion cracking, making it less suitable for wet or marine environments unless properly coated. The tradeoff is that 7075 offers substantially higher tensile strength—503 MPa vs. 276 MPa for 6061 T6.
Which is stronger, 6061 or 5052 aluminum?
6061-T6 is stronger, with a 276 MPa yield strength vs. 214 MPa for 5052-H34. However, 5052 outperforms 6061 in corrosion resistance, especially in marine environments. Choose based on whether load-bearing capacity or environmental durability takes priority.


