
Introduction
Two steel parts with identical chemistry can perform worlds apart—the difference comes down to heat treatment. Outcomes vary dramatically based on process type, temperature precision, hold time, and cooling method, and a single misstep in any variable can produce brittle, soft, or distorted results.
This guide covers what heat treatment does to metal at a microstructural level, the four primary methods used to strengthen metal, a step-by-step process walkthrough, critical control variables, and common failure points to avoid.
TL;DR
- Heat treatment strengthens metal by altering its internal crystalline structure—shape and dimensions stay unchanged
- The four core strengthening methods are hardening/quenching, tempering, normalizing, and case hardening—each targets a different mechanical goal
- Temperature accuracy, soak time, and cooling rate are the three variables that make or break any heat treatment outcome
- Carbon content, alloy composition, and part geometry all determine which process fits a given application
- In extreme wear or corrosion environments, diffusion-based surface treatments like boronizing extend service life beyond what heat treatment alone can achieve
What Does Heat Treatment Actually Do to Metal?
Metals are made of crystalline microstructures called grains. The size, arrangement, and phase of these grains govern mechanical properties like hardness, toughness, and ductility. Heat treatment manipulates grain behavior without melting the metal.
Two Primary Mechanisms
Martensite transformation occurs when rapid cooling traps carbon atoms in the crystal lattice, creating internal stress and extreme hardness. Steel quenched from high temperature doesn't give carbon atoms time to migrate—they freeze in a strained body-centered tetragonal (BCT) structure called martensite.
Diffusion, by contrast, relies on slow, controlled cooling that lets atoms migrate and form new, more stable phases. This produces microstructures like pearlite or bainite—tougher but less hard than martensite.
Composition Matters
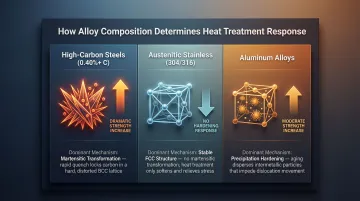
Which mechanism dominates depends entirely on what the metal is made of. Results vary significantly by alloy composition:
- High-carbon steels (above 0.40% C) harden dramatically when quenched
- Austenitic stainless steels (304, 316) may soften because they don't undergo the necessary phase transformation—their FCC austenite structure remains stable at room temperature
- Aluminum alloys respond through precipitation hardening, a different mechanism entirely
The iron-carbon phase diagram maps exactly where these phase boundaries fall—useful when specifying treatment temperatures for a given steel grade or carbon content.
The 4 Types of Heat Treatment That Strengthen Metal
"Heat treatment" is an umbrella term. The four processes below target different mechanical outcomes and are selected based on application requirements.
Hardening and Quenching
Hardening involves heating steel above its upper critical temperature—typically 815–900°C (1500–1650°F) depending on alloy and carbon content—until it reaches the austenite phase. The part is then rapidly cooled (quenched) in oil, water, brine, or forced air. Rapid cooling produces martensite, a hard, brittle crystalline structure.
Hardening maximizes surface and through-hardness but increases brittleness in the process—which is why tempering almost always follows as the next step.
Typical Hardness Ranges (HRC):
| Steel Grade | Carbon Content | As-Quenched Hardness |
|---|---|---|
| 1018 | 0.14–0.20% | 28–42 HRC (thin case only) |
| 4140 | 0.38–0.43% | 55–57 HRC |
| 4340 | 0.38–0.43% | 57–59 HRC |
| D2 | 1.40–1.60% | 63–65 HRC |
Tempering
Tempering is performed immediately after quenching. The steel is reheated to a lower temperature—typically 200–600°C (392–1112°F)—to reduce brittleness while retaining most of the hardness gained from quenching.
Tempering temperature determines the final balance between hardness and toughness. Higher tempering temperatures sacrifice more hardness but deliver greater toughness.
Historically, the visible oxide layer that forms during tempering served as a visual indicator of temperature:
| Oxide Color | Temperature (°F) | Temperature (°C) |
|---|---|---|
| Pale Yellow | 420–430 | 216–221 |
| Straw Yellow | 460 | 238 |
| Brown | 490–500 | 254–260 |
| Purple | 530–540 | 277–282 |
| Dark Blue | 570 | 299 |
These colors are approximations affected by surface finish and alloy composition, but they remain a practical reference for manual operations.
Normalizing
Normalizing involves heating steel above its upper critical temperature and then cooling it in still air (not quenched). This produces a uniform, fine-grained pearlite microstructure that increases strength and toughness while reducing internal stresses caused by forming, welding, or machining.
Normalizing produces a harder, stronger result than full annealing because faster air cooling (1–10°C/s) prevents coarse pearlite from developing. It's often used as a preparatory step before further heat treatment.
| Parameter | Normalizing | Full Annealing |
|---|---|---|
| Heating Temperature | 850–950°C | 750–830°C |
| Cooling Method | Still Air | Furnace Cool |
| Microstructure | Fine Pearlite + Ferrite | Coarse Pearlite + Ferrite |
| Primary Use | Grain refinement, stress relief | Maximum softening for machining |
Case Hardening
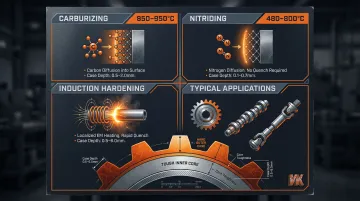
Case hardening creates a hard outer shell (the "case") while the core remains tough and ductile. Common methods include:
- Carburizing: Carbon diffusion at 850–950°C into low-carbon steel surfaces
- Nitriding: Nitrogen diffusion at 480–800°C, forming a hard compound layer without quenching
- Induction Hardening: Localized surface heating via electromagnetic induction, followed by quenching
Case hardening is the go-to choice for components that need wear-resistant surfaces but must absorb impact without fracturing—gears, camshafts, drive shafts, and pump components. Case depth (typically 0.020–0.250 inches) must be matched to application load requirements. Low-carbon alloy steels like 8620 (0.20% C) and 9310 (0.10% C) are favored base materials because they produce a hard case (60+ HRC) over a tough, ductile core.
How to Strengthen Metal with Heat Treatment: Step-by-Step
Regardless of method chosen, every successful heat treatment follows the same four-stage sequence. Skipping or rushing any stage compromises the final outcome.
Step 1: Material Verification and Preparation
Confirm the metal grade and carbon/alloy content before selecting a process. The wrong treatment for a given alloy can result in cracking, distortion, or no measurable improvement. Reference material certifications and mill test reports as the starting point.
Inspect and clean the workpiece: Surface contaminants—oils, scale, coatings—cause uneven heating, decarburization, or surface defects. Check for existing stresses, cracks, or dimensional tolerances that may be affected by thermal expansion.
Step 2: Heating to Target Temperature
Load the part into the furnace and ramp temperature at a controlled rate. Larger cross-sections require slower heating rates to prevent thermal shock and cracking—typically 20 minutes per inch of cross-section thickness for round bars.
Control the furnace atmosphere: In most hardening applications, use an inert or controlled atmosphere—nitrogen, argon, or endothermic gas—to prevent oxidation and decarburization. Decarburization strips carbon from the surface layer, directly weakening the zone you're trying to harden.
Step 3: Soaking at Temperature
Hold the part at target temperature long enough for heat to fully penetrate through the cross-section. This is the "soak time," typically calculated at one hour per inch of cross-section thickness—verify against alloy-specific charts before committing to a schedule.
Too little soak time leaves some areas in the wrong phase; too much causes grain growth. Either extreme reduces mechanical performance.
Step 4: Controlled Cooling (Quenching or Air Cooling)
Cooling medium selection:
| Quench Medium | Cooling Severity | Typical Applications | Cracking Risk |
|---|---|---|---|
| Brine (Salt Water) | Highest | Shallow-hardening plain carbon steels | Very High |
| Water | High | Low-carbon steels | High |
| Aqueous Polymer (PAG) | Adjustable (1-12% concentration) | Medium-carbon, alloy steels | Moderate |
| Oil | Moderate | Deep-hardening alloy steels (4140, 4340) | Low |
| Forced Air / Vacuum | Lowest | High-alloy tool steels (D2) | Very Low |
Match the medium to the alloy's hardenability. Water and brine cool fastest but risk cracking in high-alloy or high-carbon steels. Oil is the standard for most tool and alloy steels.
Post-Quench Operations: Quenching is only half the equation—tempering or stress relief must follow without delay. Parts remaining in the as-quenched martensitic state face high risk of stress cracking, especially in complex geometries. Begin tempering once the part reaches 52–65°C to prevent delayed cracking.
Key Parameters That Determine Heat Treatment Results
Even with the right process selected, small deviations in four control variables are responsible for the majority of heat treatment failures.
Temperature Accuracy
The transformation from one crystalline phase to another only occurs within a specific temperature window. Too low and transformation is incomplete; too high and excessive grain growth reduces toughness.
AMS 2750H, the aerospace pyrometry standard, defines how tight those windows must be:
- Class 1: ±5.0°F (±3.0°C)
- Class 2: ±10.0°F (±6.0°C)
- Class 3: ±15.0°F (±8.0°C)
Aerospace applications demand Class 1 or Class 2 tolerances. General industrial heat treating often operates at Class 3 or Class 5 (±25°F), but tighter tolerances eliminate incomplete transformations and reduce scrap. Facilities processing aerospace or critical-service components — including diffusion coating operations like VaporKote — use furnaces with digital microprocessor controls specifically to hold these tolerances consistently.
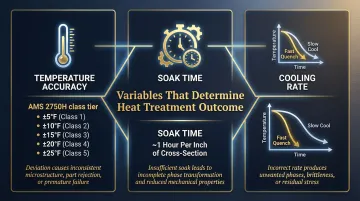
Soak Time
Soak time determines whether phase transformation is complete and uniform through the full cross-section. Inadequate soak time leaves a gradient of microstructures from surface to core — the surface may reach target hardness while the core remains soft.
Soak time must be calibrated to part size, geometry, furnace load density, and the specific alloy. A common starting rule is approximately one hour per inch of cross-section, though alloy and furnace type shift this baseline. A one-size-fits-all approach is one of the most common sources of inconsistent hardness results.
Cooling Rate
Cooling rate controls which microstructure forms:
- Faster cooling → martensite (hard, brittle)
- Slower cooling → pearlite or bainite (tougher, less hard)
The rate must match the alloy's TTT (time-temperature-transformation) curve. Deviate from it and you get the wrong microstructure — regardless of how well the heating phase was controlled.
Uneven cooling compounds the problem. Thin sections cool faster than thick sections within the same part, creating warping or internal cracking. Proper fixturing and orientation in the quench bath reduce this risk.
Alloy Composition and Carbon Content
Carbon content determines hardenability. Low-carbon steels (below ~0.25% C) are generally not hardenable by quenching, while high-carbon and alloy steels respond strongly. Alloying elements — chromium, molybdenum, nickel — shift transformation temperatures and slow critical cooling rates, which is why 4140 and 4340 steels behave so differently from plain 1018.
Not knowing or misidentifying the alloy grade is one of the most common causes of unexpected outcomes. Always match treatment parameters to the material certification data (mill certs or material test reports) before processing.
Common Mistakes to Avoid—and When to Consider Advanced Surface Treatment
Skipping or Rushing the Soaking Stage
Parts pulled from the furnace before full thermal equilibrium is reached will have inconsistent hardness across their cross-section. Always calculate soak time based on the thickest cross-section, not overall part weight.
Using the Wrong Quench Medium for the Alloy
High-alloy or high-carbon steels quenched in water can crack almost immediately. Always consult alloy-specific quench media recommendations. Never assume oil or water is universally safe.
Neglecting Post-Quench Tempering
As-quenched martensite is too brittle for most functional applications. Skipping or delaying tempering after quenching is a leading cause of premature part cracking under service stress.
Recognizing When Heat Treatment Alone Isn't Enough
Sometimes the mistake isn't in how heat treatment is applied—it's applying it where it can't solve the underlying problem. For components operating in extreme abrasion, erosion, or high-temperature corrosion environments—common in oil refining, mining, and petrochemical processing—traditional heat treatment may extend part life but cannot match the surface hardness achievable through diffusion coating processes like boronizing.
Boronizing achieves 1450–5000 HV surface hardness, far exceeding carburizing (700–850 HV). VaporKote's chemical vapor deposition process produces an intermetallic surface compound reaching 1500 Knoop hardness (RC75+ equivalent), which exceeds the hardness of tungsten carbide cutting tools. For applications where surface protection determines service life, that difference is measurable in months of additional uptime.
In abrasive wear tests, boronized steels show up to 10-fold lower material loss compared to nitriding or carburizing in erosive environments. VaporKote's boronizing process is well-suited for components that see the highest wear exposure in oil refining, mining, and petrochemical operations:

- Pump wear rings and impellers
- Valve components and seats
- High-temperature nozzles
- Extrusion and forming tooling
Frequently Asked Questions
What are the 4 types of heat treatment?
The four core types are:
- Hardening/quenching — maximizes hardness by forming martensite
- Tempering — reduces brittleness while retaining hardness
- Normalizing — produces uniform grain structure and relieves stress
- Case hardening — creates a hard surface over a tough core
The right choice depends on the metal type and application requirements.
What temperature do you heat treat steel at?
Temperature depends on the process and steel grade. Hardening typically requires heating above the upper critical temperature—roughly 815–900°C (1500–1650°F) for most carbon steels. Tempering occurs at much lower temperatures, 200–600°C (392–1112°F). Always reference alloy-specific data charts for precise temperatures.
What steel can be heat-treated?
Medium and high-carbon steels, alloy steels (like 4140, 4340), tool steels (D2, H13), and precipitation-hardening stainless steels (like 17-4 PH) respond well to heat treatment. Low-carbon steels (below ~0.25% C) and most austenitic stainless steels (304, 316) cannot be hardened through quenching.
What does heat treatment do to steel?
Heat treatment alters the crystalline microstructure of steel. The process can increase hardness and strength, improve toughness, relieve internal stresses, or create a wear-resistant surface over a tough core — depending on which method is applied. It doesn't change the part's shape or dimensions.
Does cold working strengthen a metal?
Yes — cold working increases strength and hardness through work hardening, which introduces dislocations into the grain structure via plastic deformation below the recrystallization temperature. It reduces ductility in the process and operates through a different mechanism than thermal heat treatment. The two can be combined for specific applications.
What is heat treating metal called?
The general process is called "heat treatment" or "heat treating." Specific sub-processes — hardening, annealing, tempering, normalizing, and quenching — are named after their outcome or method. Thermochemical diffusion processes like boronizing and aluminizing also fall under this broader category, used when extreme surface hardness or corrosion resistance is required beyond what conventional heat treatment provides.


