
Introduction
Wear rings in pumps, compressors, and rotating equipment face a compound threat: corrosive media, mechanical wear, and abrasive slurries attacking simultaneously. These components maintain tight clearances, control internal recirculation, and prevent metal-to-metal contact. Standard wear rings designed purely for mechanical wear often fail prematurely once corrosion enters the picture—triggering unplanned shutdowns and costly cascading damage.
Corrosion carries a price tag most engineers underestimate. The global cost reaches approximately $2.5 trillion annually—roughly 3.4% of global GDP. U.S. industries alone absorb enormous direct losses: oil and gas exploration ($1.4 billion), petroleum refining ($3.7 billion), and chemical processing ($1.7 billion) per year.
When a wear ring fails due to corrosion, the damage rarely stops there. Impellers, shafts, and pump casings suffer secondary damage, turning a small component failure into a full equipment overhaul.
What follows covers why conventional materials leave critical performance gaps, how diffusion coating technologies address the combined wear-corrosion threat, and which industries stand to gain the most.
What Are Wear Rings and Why Do They Fail in Corrosive Environments?
Wear rings (also called guide rings or wear bands) are renewable, close-running annular components installed in pumps, hydraulic cylinders, and rotating machinery. Their primary functions include:
- Absorbing radial and side loads during operation
- Guiding linear or rotary motion with precision
- Preventing destructive metal-to-metal contact between moving parts
- Maintaining tight clearance between the impeller and pump casing
In centrifugal pump impeller assemblies specifically, wear rings control the running clearance between the impeller outer diameter and the pump casing. This clearance directly impacts hydraulic efficiency by limiting internal recirculation—the leakage of high-pressure discharge liquid back toward the low-pressure suction region. Every time wear ring clearance doubles, a pump typically loses 3 to 5 points of volumetric efficiency, forcing the impeller to continuously re-energize the same liquid.
The Dual Threat: Wear and Corrosion
In standard service conditions, mechanical wear alone gradually degrades a wear ring through abrasion and surface contact. But in corrosive environments — acids, chlorides, hydrogen sulfide (H₂S) sour gas, seawater, or aggressive process fluids — chemical attack dramatically accelerates material degradation.
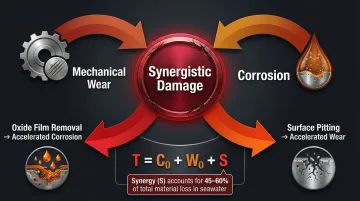
This phenomenon is called tribocorrosion: a material degradation process caused by the combined, synergistic effect of wear and corrosion. In seawater applications, high fluid velocities through tight wear ring annuli strip protective oxide films, causing wear-induced corrosion that accounts for 45% to 60% of total material loss. Total material loss (T) is expressed as T = C0 + W0 + S, where C0 is pure corrosion, W0 is pure wear, and S is the synergy between them.
The compounding effect works both ways. Corrosion undermines structural integrity through surface pitting, wall thinning, and oxidation, which accelerates the rate of mechanical wear. At the same time, mechanical wiping continuously removes protective passive oxide films, exposing bare metal to accelerated anodic dissolution (the electrochemical removal of surface material).
A pitted or oxidized surface creates stress concentration points that initiate cracking under cyclic loading. Corrosion and wear reinforce each other rather than acting independently, shortening service life far beyond what either threat would cause alone.
Common Corrosion Failure Mechanisms in Wear Rings
Identifying the specific corrosion mechanism matters because uniform corrosion rates rarely predict wear ring failure in tight-clearance environments. The primary mechanisms include:
Uniform Corrosion: Metallic wear rings exposed to acidic media without adequate protection lose material uniformly across the surface. Predictable but damaging, this mechanism reduces wall thickness and opens clearances beyond acceptable tolerance.
Crevice Corrosion: Confined spaces — such as the press-fit boundary between a wear ring and pump casing — trap stagnant fluid with limited oxygen replenishment. Oxygen depletion in these microscopic gaps prevents passive oxide film regeneration, dropping local pH and triggering aggressive acidic attack. 300-series stainless steels are highly vulnerable to this mechanism in chloride-rich environments.
Galvanic Corrosion: When dissimilar metals are electrically connected within a conductive electrolyte (such as seawater or produced water), a galvanic cell forms. The less noble material acts as the anode and corrodes rapidly. Pairing a bronze wear ring with a stainless steel impeller in seawater causes the bronze to sacrifice itself, leading to rapid clearance loss and potential ring detachment.
Erosion-Corrosion: Suspended solids in high-velocity process fluids mechanically sweep away corrosion products and passive films, continuously exposing fresh metal to attack. The narrow annular clearance of a wear ring concentrates fluid velocity, making erosion-corrosion one of the most common failure modes in slurry and produced-water service.
Stress Corrosion Cracking (SCC): Environmentally assisted cracking occurs under combined tensile stress (including residual stress from press-fitting) and specific corrosive agents. Chloride SCC affects 300-series austenitic stainless steels in aqueous chloride environments, particularly at metal temperatures above 60°C (140°F). Sulfide Stress Cracking (SSC) occurs in H₂S environments, where hydrogen atoms diffuse into high-strength, high-hardness steel lattices, causing sudden embrittlement and cracking.
The Hidden Cost of Corrosion on Industrial Wear Rings
The operational cost of corrosion-related wear ring failure extends far beyond the component replacement price. When a ring degrades, impeller clearances open up, reducing pump efficiency and flow. When the ring fails completely, the impeller, shaft, and pump casing suffer secondary damage—turning a small wear component failure into a major equipment overhaul.
Implementing available corrosion control practices could save between 15% and 35% of the estimated $2.5 trillion global annual corrosion cost, yet many facilities continue to operate with inadequate wear ring protection strategies.
The Reactive Maintenance Penalty
The hidden maintenance burden is substantial. Because corrosion often initiates in inaccessible locations and progresses unseen, many facilities don't discover wear ring failure until efficiency has already dropped significantly or a catastrophic failure forces an emergency shutdown. Relying on reactive ("run-to-failure") maintenance for pump wear rings costs 2 to 5 times more than proactive or preventive strategies due to emergency labor rates, expedited parts, and uncontrolled downtime costs.
The difference between planned replacement and reactive failure costs includes:
- Premium after-hours labor rates (often 3x standard rates)
- Expedited shipping costs for emergency parts
- Lost production during unplanned downtime
- Secondary damage to mating components (impellers, shafts, seals, bearings)
- Damage propagation to downstream equipment or piping systems

Frequency and Energy Penalties
These reactive maintenance costs compound when you account for replacement frequency. In corrosive service, standard wear rings may need replacement at a fraction of their rated service life, multiplying both direct material costs and the labor and downtime tied to repeated teardown and reassembly. A wear ring designed for 18-24 months of service may fail in 3-6 months when corrosion accelerates degradation.
Corrosion-induced clearance growth also creates ongoing energy penalties. A 3% efficiency gain achieved by restoring and maintaining tight wear ring clearances across a 25,000-horsepower pump population (typical for a 100,000-bpd refinery) decreases power usage by 4.9 million kilowatt-hours annually.
Standard Materials vs. Corrosion Resistant Wear Ring Solutions
Selecting the correct wear ring material requires balancing mechanical strength, galling resistance, and chemical compatibility—a balance that conventional materials struggle to achieve.
Traditional Wear Ring Materials and Their Limitations
Conventional wear ring materials fall into two main categories, each with critical limitations in corrosive service:
Polymer and Composite Options:
Materials like glass-filled nylon, bronze-filled PTFE, and phenolic fabric composites offer low friction and good mechanical wear resistance. They eliminate metal-to-metal galling and allow for tighter running clearances (often 50% of API minimums). However, they face significant limitations:
- Many process fluids cause swelling, degradation, or direct chemical attack
- Temperature ceilings are low: glass-filled nylon maxes out around 174°C; phenolic laminates around 90–120°C
- Compressive strength tops out at 200–350 MPa, restricting use in high-pressure applications
- Some materials absorb up to 7% water at saturation, causing dimensional instability
Metallic Options:
Carbon steel, cast iron, and bronze provide strength and dimensional stability. Without surface protection, they corrode rapidly in chloride, sulfur-containing, or acidic environments. More corrosion-resistant alloys introduce their own problems:
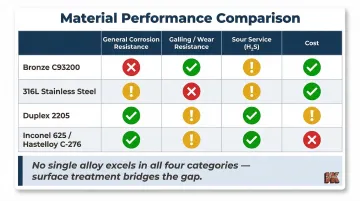
- Bronze (C93200/SAE 660): Excellent lubricity but highly susceptible to galvanic corrosion when paired with stainless steel in seawater; not recommended for strong acids or ammonia
- 316/316L Austenitic Stainless Steel: Excellent general corrosion resistance but extremely prone to galling (self-mated 316L suffers total seizure at low loads in ASTM G98 testing); highly susceptible to chloride SCC above 60°C
- Duplex Stainless Steel (2205): Superior resistance to chloride SCC and erosion-corrosion compared to 300-series, but NACE MR0175 limits standard duplex to a maximum H₂S partial pressure of 0.1 bar and maximum temperature of 232°C (450°F)
- Inconel 625 & Hastelloy C-276: Outstanding resistance to chloride SCC and sour environments but lack inherent surface hardness required to survive severe abrasive slurries without surface modification; also extremely expensive
The Core Trade-Off
Materials selected purely for corrosion resistance (high-alloy stainless steels, Hastelloy, Inconel) are expensive, often softer than needed for wear-intensive service, and may still be susceptible to localized attack. Materials selected purely for wear resistance lack the chemical inertness needed in corrosive media. In severe combined-service conditions, choosing one property over the other leaves a critical performance gap that monolithic material selection alone cannot close.
Bridging the Gap: Surface Treatment as the Solution
Surface treatment applied to a suitable base metal—rather than material substitution alone—resolves this trade-off directly. Diffusion coatings alter the substrate's surface chemistry at the atomic level, producing hardness and corrosion resistance in a single layer that no single alloy delivers at equivalent cost.
Key distinctions from conventional surface protection methods:
- Metallurgical bond: Diffusion processes integrate into the substrate rather than sitting on top, eliminating delamination risk under cyclic fatigue or impact loading
- Combined properties: A properly boronized or aluminized steel component achieves surface hardness exceeding 1500 Knoop (RC75+ equivalency) while retaining the toughness of the base metal
- No adhesion failure: Unlike electroplating or thermal spray, there is no coating-substrate interface to fail under high contact stress
How Diffusion Coatings Deliver Superior Corrosion and Wear Resistance
Diffusion coating processes—specifically boronizing and aluminizing via chemical vapor deposition—work by driving atoms of boron or aluminum into the surface of the base metal at elevated temperatures. This forms an intermetallic compound layer that is metallurgically bonded (not just adhered) to the substrate, distinguishing it fundamentally from plating or thermal spray coatings that sit on top of the surface and can delaminate.
Boronizing for Extreme Wear Protection
Boronizing is a thermo-chemical diffusion process typically performed at 700°C to 1000°C, where boron atoms diffuse into the metal lattice to form hard metal-boride compounds (FeB and Fe₂B). The process creates a "saw-tooth" morphology in carbon steels that anchors the layer to the substrate.
Extreme Surface Hardness:
VaporKote's documented surface hardness of 1500 Knoop (RC75+ equivalency) exceeds that of tungsten carbide cutting tools. The outer FeB phase reaches 1890–2340 HV (~20.9 GPa), while the inner Fe₂B phase achieves 1290–1700 HV (~17.4 GPa). This hardness directly extends wear ring service life in abrasive and erosive environments such as slurry pumps, drilling equipment, and mining applications.
NACE Compliance for Sour Service:
API 610 and NACE MR0175 strictly prohibit through-hardened renewable wear rings above HRC 22 in sour (H₂S) services due to catastrophic sulfide stress cracking risks. Because boronizing creates an ultra-hard surface while the core material remains ductile and below the HRC 22 limit, boronized wear rings comply with NACE MR0175 for sour service while providing surface hardness exceeding 1600 HV.
Tribocorrosion Resistance:
In 3.5% NaCl tribocorrosion tests (wear accelerated by corrosion), boronized steels demonstrate significantly lower wear rates and friction coefficients compared to uncoated steels, as the chemical inertness of the boride layer prevents wear-accelerated corrosion. The boride surface resists many acids, chlorides, and sulfur-containing media that rapidly attack base steel or iron.
Aluminizing for High-Temperature Corrosion
Aluminizing (via pack cementation or CVD) diffuses aluminum into the surface of ferrous alloys to form Fe-Al intermetallic layers (e.g., FeAl, Fe₃Al). At elevated temperatures, these intermetallics oxidize to form a slow-growing, highly stable, and self-healing α-Al₂O₃ (alumina) scale.
In severe refinery environments (700°C–800°C with water vapor and H₂S), aluminide coatings prevent the formation of thick, spalling iron-sulfide or iron-oxide scales. CVD aluminide coatings on steels have survived 10,000+ hours at 800°C without failure, whereas uncoated steels suffer severe attack. For wear rings in high-temperature sulfidic streams common in petroleum refining and petrochemical furnace environments, aluminizing provides proven resistance to oxidation and sulfidation.
VaporKote's Process Advantage
Both boronizing and aluminizing can be tuned precisely — and that's where VaporKote's process stands apart. Rather than applying standardized formulations, VaporKote formulates powder mixes on-site, adjusting composition to match the specific corrosive media, base metal, and operating temperature of each application. This applies across petrochemical refining, oil production, mining, and other demanding environments.
The result is a coating optimized for the exact corrosive agents and thermal stresses a component will face in service. In head-to-head comparisons, VaporKoted wear rings consistently outlast both uncoated high-alloy components and parts treated with conventional surface processes — at lower total lifecycle cost.
Practical Outcomes
Diffusion-coated wear rings deliver measurable operational benefits:
- Extends time between planned replacements, reducing scheduled downtime
- Reduces catastrophic failure risk and unplanned shutdowns
- Lowers total lifecycle cost despite higher initial component price
- Shields high-value impellers, shafts, and housings from accelerated damage when rings fail
- Maintains tight clearances longer, preserving hydraulic efficiency
Industries and Applications That Demand Corrosion Resistant Wear Rings
When wear rings degrade in corrosive service, the consequences go beyond component failure — pump efficiency drops, throughput falls, and unplanned shutdowns follow. API 610 and ISO 13709 set the baseline for clearances and materials precisely because these environments leave no margin for gradual degradation.
Pump and Rotating Equipment Applications
The centrifugal pump impeller wear ring application demands precise engineering. The wear ring maintains the critical running clearance between the impeller outer diameter and the pump casing, controlling internal recirculation and hydraulic efficiency. In corrosive service—seawater lift pumps, produced water injection, chemical transfer—corrosion rapidly widens this clearance, reducing pump efficiency and throughput.
API 610 establishes strict minimum diametral clearances based on the outer diameter of the impeller wear ring to prevent seizure. For materials with high galling tendencies (like stainless steel) or for any material operating above 260°C (500°F), an additional 0.125 mm (0.005") must be added to the minimum diametral clearance — a direct penalty to volumetric efficiency.
Hard diffusion coatings keep clearances tight throughout the service interval, protecting efficiency where standard materials would corrode and widen the gap.
Harsh Industrial Environments Served
Key industrial sectors where simultaneous wear and corrosion make standard wear rings inadequate include:
- Oil & Gas Drilling and Production: Sour service H₂S creates extreme sulfide stress cracking risk. NACE-compliant core materials (< HRC 22) paired with boronized wear faces deliver the ductility and surface hardness this environment demands.
- Petroleum Refining: High-temperature sulfidic attack requires aluminizing to form protective Al₂O₃ scales, preventing iron-sulfide scaling at temperatures up to 800°C.
- Petrochemical Processing: Process acids and caustics combined with mechanical stress require both chemical inertness and surface hardness — boronized wear rings provide both.
- Mining: Abrasive slurry with acidic groundwater drives severe erosion-corrosion. Boronized surfaces, harder than tungsten carbide, resist particle impact while chemical inertness halts corrosion acceleration.
- Pulp & Paper: Chlorine-containing bleach plant chemicals attack standard materials rapidly. Diffusion coatings hold up in these oxidizing environments where 316SS fails.
- Marine and Offshore: Seawater service combines galvanic corrosion, chloride SCC, and erosion-corrosion. Diffusion-coated alternatives to 316SS eliminate the compounding failure mechanisms standard materials cannot withstand.

VaporKote has applied boronizing and aluminizing solutions to wear ring components across these sectors since 1987, working to ASTM, ASME, API, and SAE standards — with the metallurgical documentation to support it.
Signs Your Wear Rings Are Failing Due to Corrosion
Operators must catch wear ring degradation early — before it cascades into catastrophic failure.
Performance-Based Warning Signs
These operational indicators often appear before any visual evidence of failure is accessible:
- Flow rate or discharge pressure drops as widening clearance gaps increase internal recirculation, pushing the pump off its performance curve
- Power consumption rises because the impeller must continuously re-pressurize recirculated fluid just to meet system demand
- Vibration and noise increase as corroded clearances erode the Lomakin Effect — the hydrodynamic bearing action that stabilizes the rotor
- Mechanical seal faces separate due to shaft deflection, causing leakage that worsens as rotor stiffness is lost
Visual and Physical Signs
When operational symptoms push you to open the pump, these are the physical signs that confirm corrosion is the culprit:
- Surface pitting creates stress concentration points that accelerate mechanical wear far beyond normal clearance loss
- Discoloration or rust staining on ring surfaces confirms active oxidation, not just mechanical degradation
- Asymmetric wear patterns point to localized corrosion rather than uniform contact — a clear signal of material incompatibility
- Ring OD or ID measurements outside tolerance indicate clearance loss that no re-installation can correct
The Decision Point
When a wear ring shows corrosion-related damage on inspection, it signals that the material selection or surface protection is inadequate for the service environment. Replacing it with an identical ring guarantees the same failure on the same timeline. The inspection is your opportunity to switch to a diffusion-coated wear ring — one engineered to resist the specific chemistry and conditions driving the corrosion.
Frequently Asked Questions
What is a wear ring and what is its function in an impeller?
A wear ring is a replaceable component fitted between the impeller and the pump casing that maintains a tight running clearance to control internal recirculation, improve hydraulic efficiency, and protect the impeller and casing from direct contact and wear.
What are the signs of a worn wear ring?
Operational signs include reduced pump flow or pressure, increased vibration or noise, and higher energy consumption. On inspection, look for dimensional loss, surface pitting, discoloration, and uneven wear patterns — indicators of corrosion damage rather than normal mechanical wear.
What causes wear rings to corrode faster in industrial environments?
Primary accelerants are chlorides (seawater, produced water), hydrogen sulfide in sour service, acidic or caustic process fluids, and elevated temperatures. Abrasive particles compound the damage by stripping away protective surface oxides and leaving fresh metal exposed to chemical attack.
Can existing wear rings be coated to improve their corrosion resistance?
Yes. Metallic wear rings can be processed with diffusion coatings such as boronizing or aluminizing, applied to either new replacement rings or refurbished components machined to spec. Both approaches deliver significantly better combined wear and corrosion resistance than uncoated rings.
What industries benefit most from using corrosion resistant wear rings?
Industries where corrosive environments combine with high mechanical demands benefit most: oil and gas, petrochemical processing, mining, pulp and paper, and marine or seawater applications. Standard wear ring materials degrade quickly under these conditions, making corrosion-resistant alternatives a practical necessity.


