
Introduction
Unplanned downtime from premature component failure carries serious financial consequences. Automotive manufacturers lose approximately $2.3 million per hour when production lines stop, while Fortune Global 500 companies collectively forfeit $1.4 trillion annually to unplanned downtime—representing 11% of their total revenues. For industries like manufacturing, oil & gas, and aerospace, inadequate surface hardness on critical metal parts translates directly to catastrophic wear failures, extended maintenance intervals, and costly emergency replacements.
Gas nitriding is a thermochemical surface treatment that has strengthened industrial components for nearly a century. Developed independently in the United States and Germany during the 1920s, this diffusion process delivers exceptional surface hardness, wear resistance, and corrosion protection without the dimensional distortion caused by quenching.
Unlike conventional hardening methods that require rapid cooling and extensive post-treatment machining, gas nitriding operates at subcritical temperatures—allowing components to be finish-machined before heat treatment.
This guide covers the gas nitriding process from preparation through completion, key performance benefits, compatible materials, industry applications, and how it compares with alternative surface hardening methods.
TLDR
- Gas nitriding diffuses nitrogen into ferrous alloy surfaces using ammonia gas at 495–565°C (925–1050°F)
- No quenching required, resulting in minimal distortion—ideal for precision components with tight tolerances
- Boosts surface hardness to 600–1100 HV and extends fatigue life by 80–100%
- Improves wear resistance and corrosion resistance without dimensional changes
- Works best on alloy steels containing aluminum, chromium, molybdenum, and vanadium
- Applications span gears, crankshafts, extrusion dies, aerospace components, and oil & gas equipment
What Is Gas Nitriding?
Gas nitriding is a thermochemical case-hardening process where nitrogen is introduced into the surface layer of ferrous alloys by holding the metal in an ammonia-rich atmosphere. This is a diffusion process, not a surface coating—the hardened layer is metallurgically bonded to the base metal and cannot peel or delaminate under load.
The process has existed for nearly a century. Adolph Machlet at the American Gas Furnace Company accidentally discovered it in 1906 while attempting to prevent oxidation using ammonia, filing a patent in 1908. Independently, Dr. Adolf Fry at Germany's Krupp works researched the critical role of alloying elements, filing his patent in 1921.
Modern computer-controlled atmosphere management has since transformed gas nitriding into a highly precise, repeatable process—one where case depth and surface hardness can be targeted within tight tolerances.
The defining differentiator from conventional hardening and carburizing is the absence of a quenching step. Because gas nitriding operates below the phase-transformation temperature of the alloy, components maintain dimensional stability and require minimal post-treatment finishing.
How the Gas Nitriding Process Works
Preparing the Workpiece
All hardenable steels must be hardened and tempered before gas nitriding begins. The tempering temperature must exceed the maximum nitriding temperature by at least 30°C (50°F) to prevent microstructural changes during the nitriding cycle.
Surface preparation is critical:
- Parts must be thoroughly cleaned through vapor degreasing or abrasive cleaning with aluminum oxide or silicon carbide grit
- Surfaces contaminated by buffing, burnishing, or cutting fluids inhibit nitrogen penetration and cause uneven case depth
- Any residual oils or surface films must be completely removed before loading
The Nitriding Cycle
Furnace Purging: Before temperatures exceed 150°C (300°F), air must be removed from the retort using nitrogen or ammonia purge gas. This step prevents oxidation and eliminates the explosive air-ammonia mixtures that form above 150°C.
Core Mechanism: Ammonia (NH₃) dissociates on contact with heated steel at 495–565°C, releasing nascent nitrogen that diffuses into the metal surface. This nitrogen reacts with nitride-forming elements—aluminum, chromium, molybdenum, vanadium, and tungsten—to form stable nitrides that create the hardened case.
Single-Stage vs. Double-Stage Nitriding:
| Process Type | Temperature | Ammonia Dissociation | Result |
|---|---|---|---|
| Single-Stage | 495–525°C (925–975°F) | 15–30% | Harder, thinner compound layer; initial phase runs 4–10 hours |
| Double-Stage (Floe) | 550–565°C (1025–1050°F) in second phase | 65–80% in second phase | Increased case depth, slightly reduced surface hardness, minimized brittle white layer |
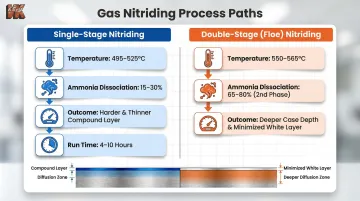
The choice depends on end-use requirements. Single-stage delivers maximum surface hardness for wear resistance, while double-stage provides deeper diffusion for high-impact applications where a brittle surface layer would be damaging.
Managing Distortion and Dimensional Changes
Three sources contribute to nitriding-related distortion:
- Relief of residual stresses from prior machining operations
- Stress introduced during the nitriding cycle itself
- Volume expansion in the hardened case layer
Despite these factors, gas nitriding produces significantly less distortion than carburizing or quench-hardening. A study analyzing 16MnCr5 gear wheels found that low-pressure carburizing followed by high-pressure gas quenching resulted in tooth thickness distortions of 0.070–0.120 mm.
Gas nitriding avoids the phase transformations that drive those dimensional changes, making it the preferred choice for precision components where tight tolerances must be maintained through heat treatment.
Stabilizing Treatment for Precision Components: For parts requiring exceptional dimensional accuracy:
- Rough grind to near-final dimensions
- Heat to 565°C (1050°F) for one hour to relieve residual stresses
- Finish grind to final tolerances
- Proceed with gas nitriding
Components not ground after nitriding typically exhibit excellent inherent dimensional stability, allowing finish-machining before heat treatment and eliminating costly post-hardening grinding.
Key Benefits of Gas Nitriding
Surface Hardness by Steel Grade
Nitrided steels achieve high case hardness that varies by steel grade and alloying elements:
- Nitralloy grades (aluminum-containing): >60 HRC (1110 HV)
- 4140 chromium-molybdenum steel: 58–65 HRC (600–800 HV)
- H13 hot work tool steel: 1000–1100 HV
- M2 high-speed steel: 62–67 HRC (1000–1200 HV)

A critical advantage: nitrided cases maintain hardness at elevated temperatures up to the nitriding temperature (~500–600°C), unlike carburized cases that soften at much lower temperatures.
Wear Resistance and Anti-Galling
The compound zone (white layer) at the surface consists primarily of epsilon (ε-Fe₂₋₃N) and gamma prime (γ'-Fe₄N) iron nitrides. This ceramic-like layer provides excellent sliding wear resistance, anti-galling properties, and a low coefficient of friction. Because this is a diffusion zone rather than a coating, there is no risk of delamination under cyclic or impact loading.
Fatigue Life Improvement
Nitrogen diffusion creates compressive residual stresses in the case that counteract tensile stresses responsible for fatigue crack initiation. Research on gas-nitrided 4140 steel demonstrates that while smooth axial fatigue strength improves by approximately 20%, the improvement for notched components is dramatic—high-cycle fatigue strength increases from 80% for blunt notches to more than 100% for sharp notches representative of gear tooth roots. Rotating bending fatigue tests on SCM440 steel showed gas nitriding increased the fatigue limit from 750 MPa to 1050 MPa.
Corrosion Resistance
The compound zone increases resistance to corrosive environments. A controlled post-oxidation step can further enhance this property by transforming the surface into a 1–3 μm magnetite (Fe₃O₄) film. In ASTM B117 salt spray testing, nitrided and post-oxidized carbon and low-alloy steels achieve up to 400 hours of protection with minimal corrosion—far surpassing untreated materials that fail within 24–48 hours. That 16× improvement in corrosion life makes it a practical choice for outdoor equipment, fluid-handling components, and assemblies exposed to mild chemicals.
Economic Advantage
Because gas nitriding requires no quenching, components can be finished to final dimensions before treatment. This eliminates expensive post-heat-treat hard grinding, reduces scrap rates from distortion, and shortens manufacturing cycle times. The total cost of ownership is measurably lower than carburizing or through-hardening processes that require post-treatment machining to correct quench-induced distortion.
Which Materials Are Best Suited for Gas Nitriding
Steel composition is the critical variable. Steels containing nitride-forming elements—aluminum, chromium, molybdenum, vanadium, and tungsten—yield superior results by forming nanoscale precipitates (AlN, CrN, VN) within the ferrite matrix. These precipitates provide strong precipitation strengthening and thermal stability.
Primary Steel Categories for Gas Nitriding
Aluminum-Containing Low-Alloy Steels (Nitralloy series):
- Nitralloy 135M, N, and EZ grades contain approximately 1.2% aluminum
- Achieve maximum surface hardness >60 HRC (1110 HV)
- Trade-off: limited ductility in the case layer
- Best for applications requiring extreme wear resistance
Medium-Carbon Chromium-Containing Steels:
- 4100, 4300, 5100, 8600 series
- Achieve 58–65 HRC (600–800 HV) surface hardness
- Better ductility than aluminum-bearing steels, making them the practical choice for high-stress structural components
Hot-Work Die Steels:
- H11, H12, H13 grades
- Achieve ~1000–1100 HV surface hardness
- Excellent for extrusion dies and forging tools
- Retain hardness at elevated operating temperatures
Air-Hardening Tool Steels:
- A-2, D-2, S-7 grades
- High wear resistance for stamping and forming dies
- Good dimensional stability during nitriding
High-Speed Steels:
- M-2, M-4 grades
- Achieve 62–67 HRC (1000–1200 HV)
- Excellent for cutting tools and precision components
Select Stainless Steel Grades:
- 304, 410, 420, 17-4 PH
- Require special depassivation steps to remove the chromium-oxide layer
- Gas nitriding struggles with passive oxide removal; plasma nitriding is often preferred
Plain Carbon Steels—Generally Unsuitable:
Plain carbon steels lack strong nitride formers, producing brittle, spalling cases with minimal hardness improvement in the diffusion zone. The resulting iron-nitride compound layer lacks subsurface support and is prone to delamination under load.
Gas Nitriding Applications Across Industries
Gas nitriding's combination of high surface hardness, minimal distortion, and corrosion resistance makes it invaluable wherever components face demanding wear and fatigue conditions. It is especially valuable for parts with complex geometries or tight tolerances where post-treatment machining must be minimized.
Automotive and Heavy Equipment
Common components treated include gears, crankshafts, camshafts, cam followers, and valve components.
Gas nitriding extends drivetrain component life by improving both fatigue resistance and wear properties. A case study involving 4140 steel gears for wind turbines showed that adding gas nitriding to a quench-and-temper cycle increased wear life by over 400% while eliminating post-treatment distortion.

That fatigue life improvement directly reduces maintenance intervals and prevents catastrophic failures in high-stress powertrain applications.
Aerospace and Defense
Gas nitriding is specified under SAE standards AMS 2759/6 and AMS 2759/10 for aerospace applications where tight dimensional tolerances and high fatigue strength are non-negotiable. The computer-controlled process delivers consistent, repeatable results across production batches — and minimal distortion means components meet strict dimensional requirements without extensive post-treatment machining.
Typical aerospace components treated include:
- Precision structural components
- Landing gear parts
- Actuator components
- Firearm components
Oil & Gas, Petrochemical, and Mining
In harsh field environments, corrosion and wear resistance directly reduce costly unplanned downtime. H13 hot work tool steel dies used for aluminum extrusion, for example, are routinely gas nitrided to prevent abrasive wear and aluminum soldering to the die bearing. A post-oxidation step adds further corrosion protection for parts exposed to aggressive process fluids.
Common applications include extruder screws, drilling components, pump parts, and injection molding components.
Tooling and Die Applications
Gas nitriding extends tool service life by providing a hard, wear-resistant surface that holds dimensional accuracy through repeated thermal and mechanical cycling. Duplex coatings (nitriding + PVD) can push life further still, but gas nitriding is the foundational treatment that allows dies to withstand severe thermal fatigue and galling in the first place.
Tooling applications regularly treated include:
- Extrusion dies
- Forging dies
- Stamping dies
- Plastic injection mold tools
Gas Nitriding vs. Other Surface Hardening Methods
Gas Nitriding vs. Ion (Plasma) Nitriding
Both processes diffuse nitrogen into the surface, but their operational mechanics dictate different ideal use cases:
| Capability | Gas Nitriding | Plasma (Ion) Nitriding |
|---|---|---|
| Temperature Control | Excellent precision and uniformity; consistent case depths | Sensitive to part emittance and thermocouple placement; temperature variances up to 28°C (50°F) possible |
| Batch Size | Ideal for large quantities and dense loads; lower gas consumption per batch | Requires specific spacing between parts for uniform glow discharge |
| Internal Surfaces | Gas easily flows into deep blind holes and complex internal geometries | Struggles to penetrate bores smaller than 0.6–0.8 mm |
| Masking | Requires copper plating or specialized masking paint | Simple, reusable mechanical masking (steel plates, bolts) |
| Stainless Steel | Requires aggressive chemical depassivation to remove chromium-oxide layer | Ion bombardment naturally sputters and cleans passive oxide layer |
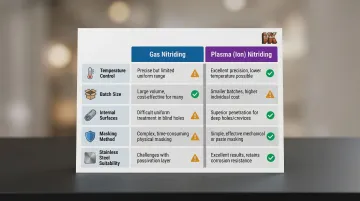
Choose gas nitriding for high-volume parts with complex internal geometries and when temperature precision is critical. Plasma nitriding is the better fit for stainless steels requiring surface activation or when selective masking of specific areas is needed.
Gas Nitriding vs. Carburizing and Quench Hardening
| Feature | Gas Nitriding | Carburizing |
|---|---|---|
| Case Depth | 0.1–0.8 mm (0.004"–0.031") | 1.0–7.5 mm (0.040"–0.300") |
| Distortion Risk | Very low; subcritical temperatures with no quenching | High; austenitizing temperatures (870–930°C) followed by rapid quenching causes significant volumetric distortion |
| Post-Machining | Minimal to none; parts finish-machined before treatment | Extensive hard-grinding required to correct quench-induced distortion |
| Material Suitability | Requires alloy steels with nitride formers | Works on carbon steels and alloy steels |
Carburizing achieves deeper case depths and works on plain carbon steels, making it suitable for applications requiring substantial hardened depth. Gas nitriding wins on dimensional stability, surface finish preservation, and reduced total manufacturing cost for precision components.
Advanced Diffusion Coatings for Extreme Hardness
For applications demanding extreme surface hardness beyond what gas nitriding can provide—such as components facing severe abrasion, high-temperature oxidation, or erosive wear in slurries—advanced diffusion coating processes like boronizing offer significantly greater surface hardness. VaporKote's boronizing process reaches 1500 Knoop (RC75+ equivalency)—harder than tungsten carbide cutting tools—making it a direct upgrade for engineers whose components fail under conditions where gas nitriding's hardness ceiling isn't enough.
Frequently Asked Questions
How does gas nitriding work?
Ammonia gas dissociates on contact with heated steel at 495–565°C, releasing nascent nitrogen that diffuses into the surface and reacts with alloying elements (aluminum, chromium, molybdenum, vanadium) to form a hard nitride case. No quenching is required, eliminating distortion.
How long does gas nitriding take?
Cycle times vary by case depth, steel grade, and whether single- or double-stage processing is used — ranging from 10–130 hours total, with the initial high-dissociation phase running 4–10 hours. A 0.5 mm case depth typically requires around 48 hours.
What is the hardness of gas nitriding process?
Surface hardness depends on steel grade. Aluminum-containing Nitralloy grades achieve the highest case hardness at >60 HRC (1110 HV), while chromium-molybdenum steels like 4140 offer 58–65 HRC (600–800 HV) with better ductility. Hot work tool steels reach 1000–1100 HV.
How much does gas nitriding cost?
Cost depends on part size, batch volume, cycle time, and steel grade. Total cost of ownership is the more useful measure — gas nitriding reduces post-treatment machining, eliminates distortion-related scrap, and extends component life. Request a quote from a qualified heat treating provider based on your specific requirements.
What is a nitride finish?
A nitride finish refers to the hardened surface layer produced by the nitriding process, consisting of a compound zone (white layer of iron nitrides) and a diffusion zone beneath containing precipitated alloy nitrides. It is not a coating but an integral part of the metal surface, providing improved hardness, wear resistance, and corrosion resistance without risk of delamination.


