
Introduction
Process hardening encompasses a category of heat treatment methods used to increase the hardness, wear resistance, and load-bearing capacity of metal components. Unlike general heat treatment—which includes softening, stress relief, and grain refinement—process hardening targets resistance to plastic deformation and wear through controlled microstructural transformation.
This guide is written for engineers, maintenance professionals, and procurement teams in manufacturing, oil & gas, petrochemical, mining, and aerospace — anyone specifying hardening processes for components under demanding service conditions.
The cost of getting it wrong is steep. When a mining haul truck fails, downtime runs $5,000–$20,000 per hour, with major breakdowns reaching $2 million per day in lost production. Across industrial sectors, unplanned downtime costs manufacturers an estimated $50 billion annually, with median per-incident costs exceeding $125,000 per hour.
We'll cover through hardening, surface hardening, diffusion coatings, and non-martensitic methods — along with the material and process variables that determine success or failure in service.
TL;DR
- Process hardening modifies metal microstructure to increase hardness, strength, and wear resistance
- Through hardening affects the entire part; surface hardening creates a hard outer layer while preserving a tough core
- Carbon content, alloy composition, part geometry, and operating environment drive method selection
- Quenching is the cooling step; hardening is the complete process — don't use them interchangeably
- Tempering follows quenching to restore toughness and reduce brittleness without fully reversing hardness gains
What Is Process Hardening?
Process hardening is a controlled heat treatment technique applied to metals—primarily steels—to increase resistance to plastic deformation by modifying internal microstructure. Depending on the method, this modification occurs through the full cross-section or selectively at the surface. The result: higher yield strength, surface hardness, fatigue resistance, and wear resistance. A hardened part holds its geometry and function longer under load, friction, and impact.
Unlike annealing, which softens metal and relieves stress, or normalizing, which refines grain structure, hardening specifically targets resistance to deformation and wear. Controlled heating followed by rapid quenching drives the formation of martensite—the hard, body-centered tetragonal phase responsible for the dramatic increase in hardness. The specific method chosen determines how deep that transformation reaches and which mechanical properties are prioritized.
Main Heat Treating Methods Used in Industry
Process hardening divides into two parent categories: through hardening (full-section transformation) and surface/case hardening (surface-layer transformation with softer core). This distinction matters because engineers must match the hardening method to the part's structural requirements—whether the entire cross-section needs high strength or only the surface requires wear protection.
Through Hardening: Quenching and Tempering
Steel is heated above its critical temperature until it fully austenitizes, then rapidly quenched to form martensite throughout the cross-section. Tempering follows to reduce brittleness and restore toughness—quenching alone without tempering is not a finished process. While as-quenched martensitic steels can reach extreme hardness (up to 60 HRC), they suffer from severe quench embrittlement and intergranular fracture risk if put directly into service. Low-temperature tempering (150–200°C) is mandatory to restore ductility and achieve a balanced, fracture-resistant component.
Quench media severity:
- Air (H-value 0.02): Very slow cooling for highly alloyed air-hardening tool steels like D2
- Oil (H-value 0.25–1.1): Moderate cooling that minimizes distortion and quench cracking in alloy steels like 4140 and 4340
- Water (H-value 0.9–4.0): Severe cooling for low-hardenability carbon steels, but poses high cracking risk
- Brine (H-value 2.0–5.0): Most severe cooling; rapidly breaks the vapor blanket stage
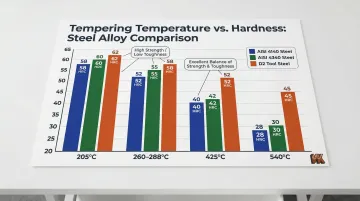
Tempering outcomes for common alloys:
- AISI 4140: Tempering at 205°C yields 52–57 HRC (high strength, low toughness); tempering at 540°C yields 28–32 HRC (excellent balance for shafts)
- AISI 4340: Tempering at 425°C yields ~46 HRC; tempering at 540°C yields ~39 HRC
- D2 Tool Steel: Tempering at 260–288°C yields 58–60 HRC (optimal wear/toughness balance)
Surface Hardening: Carburizing and Nitriding
Low-carbon steels (typically ≤0.25% C, such as 1018 or 8620) lack the carbon necessary to form fully martensitic structures through standard quenching. Carburizing diffuses carbon into low-carbon steel surfaces at high temperatures (843–1038°C), followed by quenching to form martensite. Nitriding diffuses nitrogen at lower temperatures (491–566°C), forming hard nitride compounds without requiring a quench step—eliminating distortion risk.
Process comparison:
| Feature | Carburizing | Nitriding |
|---|---|---|
| Temperature | 843–1038°C | 491–566°C |
| Quench required | Yes (oil/polymer) | No (slow cooling) |
| Case depth | 0.5–3.0+ mm | 0.1–0.88 mm |
| Surface hardness | 58–65 HRC | Up to 92.5 HR15N (~65–72 HRC equivalent) |
| Suitable alloys | 8620, 9310, 5120, 1018 | Nitralloy 135, 4140, 4340, H11, H13 |
Gas nitriding operates below tempering temperatures and eliminates the quench step entirely, achieving surface hardnesses up to 92.5 HR15N on specialized alloys like Nitralloy 135. This makes nitriding ideal for complex, tight-tolerance components where post-heat-treat machining is cost-prohibitive.
Surface Hardening: Induction and Flame Hardening
Induction hardening uses electromagnetic heating to rapidly heat only the surface, followed by immediate quenching. Flame hardening applies direct flame to the surface. Both create hard, wear-resistant surfaces while maintaining the original ductile core. These methods require steel with sufficient carbon (minimum 0.35–0.40% C) to form martensite, and are typically selected for localized hardening of large or complex parts like gears, shafts, and cams.
Frequency dictates depth in induction hardening:
| Frequency | Range | Case Depth | Applications |
|---|---|---|---|
| Low (LF) | 1–10 kHz | 5.0–20.0 mm | Large rollers, heavy axles, deep-case crankshafts |
| Medium (MF) | 10–100 kHz | 1.5–6.0 mm | Automotive shafts, spindles, camshaft lobes |
| High (HF) | 100–500 kHz | 0.5–2.0 mm | Small gear teeth, pins, thin-walled components |
The machine's frequency inversely controls current penetration depth. High frequencies yield shallow cases; low frequencies yield deep cases. AISI 1045 (0.45% C) is the most common choice, achieving 45–55 HRC. AISI 4140 (0.40% C) is used for higher-stress applications, achieving 50–60 HRC.
Diffusion Coatings: Boronizing and Aluminizing
Where induction and flame hardening rely on rapid thermal cycles, diffusion coatings take a chemistry-first approach — driving elemental atoms into the base metal surface at elevated temperatures to form intermetallic compounds. The resulting hardness levels exceed what conventional heat treatment can achieve.
Boronizing diffuses boron atoms into the metal lattice at 800–1050°C, forming ultra-hard iron boride phases (FeB and Fe₂B). The FeB phase reaches approximately 2000 HV; Fe₂B reaches 1200–1700 HV. Case depths typically range from 20 µm to 100 µm.
VaporKote's CVD boronizing process achieves 1500 Knoop surface hardness (RC75+ equivalency), outperforming standard carburized or nitrided surfaces in extreme wear and corrosion environments across petrochemical, mining, and oil & gas applications.
Aluminizing (calorizing) diffuses aluminum into steel and superalloy surfaces to form intermetallic phases (FeAl, Fe₃Al, Fe₂Al₅). Performed via pack cementation (ASTM B875) or gas-phase aluminizing at 600–800°C, the process generates a dense Al₂O₃ (alumina) scale that resists high-temperature oxidation and sulfidation — the primary degradation modes in petrochemical and power generation environments.
| Process | Primary Phases | Hardness | Primary Benefit |
|---|---|---|---|
| Boronizing | FeB, Fe₂B | 1200–2000 HV | Extreme abrasive wear resistance; low friction coefficient |
| Aluminizing | FeAl, Fe₃Al, Fe₂Al₅ | 600–1000 HV | High-temperature oxidation and sulfidation resistance |

Work Hardening and Precipitation Hardening
Work hardening (strain hardening) strengthens ductile metals through plastic deformation. As the metal deforms, dislocation density increases dramatically — the repulsive strain fields between tightly packed dislocations hinder further dislocation motion, increasing hardness and yield strength while reducing ductility.
Common alloys include austenitic stainless steels (304) and Hadfield manganese steel, which rapidly work-harden under heavy impact — making them the standard choice for mining crusher jaws and railway components.
Precipitation hardening (age hardening) builds yield strength by forming fine, uniformly dispersed precipitates within the metal matrix. The three-stage process:
- Solution treatment — heat to dissolve alloying elements into a single phase
- Quench — rapid cooling traps solutes in supersaturated solid solution
- Aging — controlled reheating below the solvus temperature allows coherent precipitates to form, creating lattice strains that block dislocation motion
Common alloys include 17-4 PH stainless steel and aerospace aluminum alloys like 2024 and 7075. Neither precipitation hardening nor work hardening produces martensite, which makes them the right choice when the application demands corrosion resistance or high impact toughness that carbon steels cannot deliver.
How Process Hardening Works: The Core Steps
The general principle underlying all thermal hardening methods: controlled heating drives microstructural transformation —for example, ferrite to austenite in steel—, and the cooling rate or diffusion medium determines the final phase and hardness level. Faster cooling traps carbon in solution and forms martensite; slower cooling allows softer pearlite or bainite to form. Each step in the process builds on this principle.
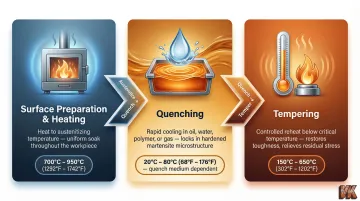
Step 1: Surface Preparation and Pre-Treatment
Parts must be cleaned of oils, oxides, and scale before hardening. Some methods require pre-machining to final dimensions; others allow post-hardening finishing. Surface contamination leads to uneven diffusion or quench cracking. Clean surfaces ensure uniform heat transfer and consistent diffusion during the transformation cycle.
Step 2: Heating and Transformation
The part is heated to the method-specific temperature—austenitizing range for quench hardening, diffusion temperature for boronizing or carburizing. Soak time (how long the part is held at temperature) controls case depth or through-thickness transformation (hardening the part's full cross-section). Temperature uniformity is critical; this is where furnace quality directly affects outcome.
Non-uniform heating produces soft spots, dimensional distortion, and hardness gradients that are difficult to predict or correct downstream.
Step 3: Quenching, Cooling, and Post-Treatment
The cooling step locks in the hardened microstructure. Common quench media include:
- Oil — moderate cooling rate, good for alloy steels
- Water or brine — fast cooling, higher cracking risk
- Polymer solutions — adjustable rate between oil and water
- Gas or air — slow cooling, used for tool steels and diffusion processes
Tempering follows quench hardening to relieve residual stress and reduce brittleness without significantly sacrificing hardness. Diffusion-based processes like nitriding and boronizing typically skip the quench step entirely, which reduces distortion risk on precision components. The quench medium and cooling rate must match the steel's hardenability—cool too slowly and soft phases form; cool too rapidly and cracking occurs.
Understanding these three steps clarifies why process selection matters as much as execution—each variable compounds across the cycle.
Why Industrial Operations Rely on Process Hardening
Unhardened or under-hardened components in heavy-duty environments suffer accelerated wear, dimensional loss, unexpected failure, and high replacement costs. In mining alone, equipment runs 5,000 to 7,000 hours per year under heavy loads, abrasive dust, and temperatures ranging from -50°C to 50°C.
Proactive repairs and optimized component lifecycles cost 4 to 5 times less than emergency reactive maintenance. Predictive maintenance combined with proper component hardening can reduce maintenance costs by 18–25% and cut downtime by 30–50%.
Industries using process hardening most extensively:
| Industry | Typical Components | Governing Standards |
|---|---|---|
| Oil & gas | Drill components, valves, pump parts | API Spec 6A, 6D, 7-1 |
| Petrochemical | Heat exchanger tubes, reactor internals | ASME B16.34 |
| Mining | Wear plates, bucket teeth, crusher jaws | — |
| Aerospace | Landing gear, bearing races | — |
| Manufacturing | Gears, dies, cutting tools | AGMA 6033-C08 |

These standards aren't just paperwork. In regulated industries, specifying the wrong hardening method or incorrect case depth can produce non-compliant parts — leading to regulatory violations and field failures with serious safety and financial consequences.
Key Factors That Affect Hardening Outcomes
Several material and process variables control hardening effectiveness:
Material variables:
- Higher carbon content enables greater hardness; alloying elements improve hardenability depth
- Hardenability — the ability of steel to harden to a given depth — is distinct from hardness itself
- Part geometry and cross-section thickness affect quench uniformity and achievable case depth
Process variables:
- Surface contamination at treatment time prevents uniform diffusion
- Quench medium and cooling rate must match the steel's hardenability to achieve the target microstructure
- Soak time and temperature uniformity govern case depth and through-thickness transformation
Hardenability vs. hardness: Hardness measures resistance to indentation; hardenability measures the depth to which a steel will harden when quenched — two different properties often confused. Engineers quantify hardenability using the Jominy end-quench test (ASTM A255), where a standardized heated cylinder is water-quenched at one end and hardness is measured along its length.
Alloying elements like chromium, molybdenum, nickel, and manganese measurably increase hardenability by shifting the Continuous Cooling Transformation (CCT) curve to the right, allowing deeper martensite formation at slower cooling rates.
These material and process variables don't exist in isolation — the operating environment shapes which tradeoffs matter most.
Operating condition factors:
- Does the part face abrasive wear, corrosive attack, impact loading, or a combination?
- Is distortion tolerance tight?
- Has the part already been machined to final dimensions (which limits methods requiring post-treatment grinding)?
Common Misconceptions and When to Reconsider Process Hardening
Three frequent misunderstandings:
Quenching is only the cooling step within a hardening cycle — not the full process. Hardening includes heating, soaking, quenching, and tempering as a sequence.
Harder is not always better. Over-hardened parts become brittle and prone to cracking under impact — untempered martensite is susceptible to intergranular fracture. The optimal hardness balances wear resistance with toughness.
Not all steels harden the same way. Low-carbon steels cannot through-harden via quench alone — they require carburizing or other case hardening first, since martensite formation needs a minimum carbon content of 0.35–0.40%.
These misconceptions often lead to over-specification or misapplied methods. Knowing when hardening adds genuine value is just as important as knowing how to apply it.
When process hardening may be unnecessary or counterproductive:
- Parts operating in low-stress, low-wear environments where base material properties are sufficient
- Parts with complex geometries where quench distortion would exceed dimensional tolerances
- Dissimilar metal assemblies where differential thermal expansion creates cracking risk
Signals that a hardening method is chosen by default, not by design:
- Over-specification of case depth beyond functional requirements
- Applying through hardening to parts that only need surface protection
- Selecting a method based on equipment availability rather than material and performance criteria
Frequently Asked Questions
What is process hardening?
Process hardening is a heat treatment method applied to metals—primarily steel—to increase hardness and wear resistance by altering the material's microstructure through controlled heating and cooling or thermochemical diffusion. The process modifies internal atomic arrangements to increase resistance to plastic deformation.
What is the purpose of hardening?
Hardening increases a metal's resistance to plastic deformation, wear, and fatigue—extending the operational life of components that face friction, impact, or heavy load. Hardened parts maintain dimensional stability longer under stress, reducing maintenance costs and unplanned downtime.
What are the two types of hardening?
Through hardening hardens the full cross-section of a part by transforming the entire volume to martensite. Surface or case hardening creates a hard outer layer while preserving a tough, ductile core—providing wear resistance at the surface and impact resistance in the core.
What is the difference between hardening and quenching?
Quenching is the rapid cooling step that locks in the hardened microstructure. Hardening is the full process—heating to austenitizing temperature, soaking, quenching, and tempering—designed to achieve a target hardness. Quenching alone, without tempering, leaves steel too brittle for service use.
What is the difference between tempering and hardening?
Hardening increases brittleness as it forms martensite through rapid quenching. Tempering is a follow-on step at lower temperatures (150–650°C) that reduces internal stress, allows trapped carbon to precipitate as fine carbides, and restores toughness without fully reversing the hardness gain.
What are the 4 processes of heat treatment?
The four core heat treatment processes are annealing (softening and stress relief), normalizing (grain refinement), hardening (martensite formation), and tempering (toughness restoration after hardening). Case hardening methods such as carburizing, nitriding, boronizing, and aluminizing are specialized extensions applied when only the surface requires property changes.


